CLINICAL DATA
A 21-year-old female patient, black, from Salvador, BA.
Since her childhood, the patient was diagnosed with double outlet right ventricle (DORV) with anterior aorta, severe pulmonary valve stenosis (PVS), hypoplasia of the pulmonary trunk and pulmonary branches of preserved calibers, systemic-pulmonary collateral arteries and left modified Blalock-Taussig (BT) shunt.
The BT operation occurred at 8 years, with interruption of medical treatment up to the worsening of cyanosis and dyspnea intensified over the past two years. During this period, the patient used only aspirin.
On physical examination she was in good general condition, ruddy, hydrated, eupneic, and cyanotic 3+/4+. Double, regular heart rhythm, with hypophonetic sounds at left upper sternal border and presence of aortic systolic murmur 2+/6+. On the left sternal border presented accentuated second heart sound and on right upper sternal edge systolic murmur of 5+/6. Bilateral vesicular murmur and without adventitious sounds. Normal abdomen. Arterial pressures and normal pulses in all four limbs, with significant digital clubbing. Peripheral O
2 saturation of 84%.
ELECTROCARDIOGRAM
Sinus rhythm, heart rate of 75 beats per minute, SÂP + 60º, SÂQRS + 90º, PR interval 0.12s, QT interval 0.44s, QTc 0.49s, indicating right ventricular overload.
RADIOGRAPHY
Visceral situs solitus in levocardia. Normal heart area with a cardiothoracic index of 0.46 and thinning of the middle mediastinum region. Pulmonary vascular network within normal limits.
ECHOCARDIOGRAM
Situs solitus in levocardia, normal venoatrial, atrioventricular and ventriculoarterial connections, with DORV. Aorta positioned anterior to the pulmonary trunk, severe PVS with major pulmonary valve annulus diameter of 5 mm and systolic peak right ventricular (RV) systolic gradient - pulmonary trunk (PT) of 88mmHg.
Ostium secundum interatrial communication of 18 mm and three muscular interventricular communications, with one RV inlet and two apical. Left BT with continuous flow of 2.6 m/s and signs of restriction.
DIAGNOSIS
The worsening of clinical symptoms, increased cyanosis and polycythemia, requiring constant bloodletting for adequacy of hemoglobin levels were determining factors for the indication of surgical intervention.
Additional tests basically showed appropriate diameter of the pulmonary arteries (Figure 1) and the presence of multiple interventricular communications, which could impair the intracardiac correction of the disease.
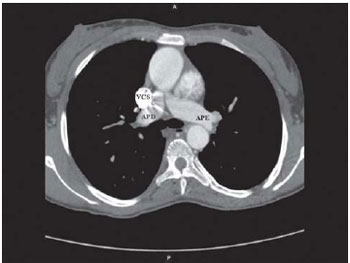
Fig. 1 - Milti-detector CT scan axial cut showing RPA with suitable diameter. RPA: right pulmonary artery, LPA: left pulmonary artery, SVC: superior vena cava
Thus, several possibilities could be used for treatment, among them: the tunneling the left ventricle into the aorta with a connection between the RV and the PT, the confection of other BT or central shunt from the aorta to the pulmonary arteries, Jatene's operation or even repair with bidirectional Glenn or Fontan-type univentricular physiology [1,2].
OPERATION
Median transsternal thoracotomy, opening of the pericardium and external analysis of cardiac structures. The aorta originated anterior to the PT, which had reduced diameter. The external anatomy of the appendages has called our attention, which were both positioned at left, featuring a juxtaposition of atrial appendages.
Dissection of TP and branches was started, as well as superior vena cava (SPV) and the azygos vein, which was ligated and sectioned. The caliber of the right pulmonary artery was near normal, which would allow the use of the univentricular physiology and guided to the bidirectional Glenn operation without cardiopulmonary bypass. Confection of pockets in the transition between the SVC and innominate vein and in the right atrial appendage (RAA) positioned at left, heparinization with 4 mg/kg and the introduction of the cannula between the VCS and the RAA (Fig. 2/video 1).
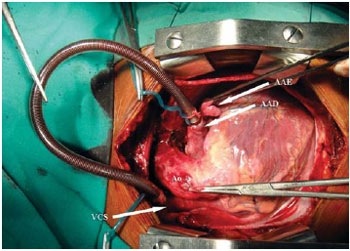
Fig. 2 - Intraoperative aspect with the cannula between the SVC and the RAA. Note the juxtaposition of atrial appendages with the RAA and the LAA at left. RAA: right atrial appendage, LAA: left atrial appendage, Ao: aorta, SVC: superior vena cava
Video 1 - Cannula between the SVC and the RAA juxtaposed to the LAA, before the confection of anastomosis between SVC and RPA. RAA: right atrial appendage, LAA: left atrial appendage, SVC: superior vena cava, RPA: right pulmonary artery.
Proximal and distal clamping in the SVC, section and suture of the proximal stump to the right atrium using 6-0 polypropylene yarn. Partial clamping of the right pulmonary artery (RPA), incision in the upper side and end-to-side anastomosis of the SVC in the RPA using polypropylene 6-0 yarn. Unclamping in the SVC and RPA after 16 minutes of using the cannula to drain the blood from the SVC.
Ligation of the pulmonary trunk with 2-0 non-absorbable yarn. Removal of the cannula between the SVC and the AAD. Reversal with protamine sulfate. Review of hemostasis, drainage of the pericardial sac and closure of the chest as usual practice.
Postoperatively, the patient evolved well, with no use of inotropic drugs, extubated early, with improvement of cyanosis, peripheral O
2 saturation of 92% and echocardiogram showing adequate drainage flow between the SVC and RPA.
It is important to note that the left BT, since it is restrictive, was not ligated, remaining as another source of pulmonary blood flow beyond the bidirectional Glenn procedure.
REFERENCES
1. Brown JW, Ruzmetov M, Okada Y, Vijay P, Turrentine MW. Surgical results in patients with double outlet right ventricle: a 20-year experience. Ann Thorac Surg. 2001;72(5):1630-5. [MedLine]
2. Croti UA, Braile DM, Godoy MF, Murillo HG, De Marchi CH, Chigutti MY, et al. A operação de Glenn bidirecional deve ser realizada com ou sem o auxílio de circulação extracorpórea? Rev Bras Cir Cardiovasc. 2004;19(3):274-9. View article
Article receive on Tuesday, February 2, 2010


 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license