Carolina Pelzer SussenbachI; João Carlos GuaragnaII; Rômulo Soares CastagninoI; Jaqueline PiccoliIII; Luciano Cabral AlbuquerqueII; Marco Antônio GoldaniI; João Batista PetraccoI; Luiz Carlos BodaneseII
DOI: 10.5935/1678-9741.20130060
ASA: Acetylsalicylic acid
UA: Unstable Angina
BB: Beta-Blocker
preop IAB: Preoperative Intra-aortic balloon
CPB: Cardiopulmonary bypass
FC: functional class
DM: Diabetes mellitus
CKD: Chronic kidney disease
LVEF: Left ventricular ejection fraction
SAH: Hypertension
CI: confidence interval
ACE: inhibitor angiotensin converting enzyme
SL LCT: Severe lesion of the left coronary trunk
P: Statistical Significance
OR: Odds Ratio
ACSST: Acute coronary syndrome without ST elevation
INTRODUCTION
Unstable angina (UA) is a leading cause of hospital admission, and their occurrence is correlated with increased mortality in both the short- and the long-term [1]. Recent studies have shown that treatment with angioplasty or coronary artery bypass graft (CABG ) have less favorable outcomes in the treatment of patients with unstable angina (UA) compared with those with stable angina. Advances in the treatment of coronary artery and the techniques tend to decrease the difference [2,3].
Treatment of UA can vary from conventional strategy to an early invasive strategy and may be indicated both surgical and percutaneous revascularization [4]. Myocardial revascularization can control the persistent ischemia and progression to acute myocardial infarction, in addition to providing symptomatic relief as well as prevent ischemic complications [5].
There are different ways to define UA. According Braunwald, UA is angina pectoris (ischemic or equivalent) with at least one of three clinical characteristics: 1) occurs at rest (or with minimal effort), usually lasting longer than twenty minutes (if not interrupted by the nitroglycerin), 2) is described as an intense and frank pain and recent onset (less than 1 month), 3) occurs in a crescendo pattern (e.g., more intense, prolonged or frequent than previously), in the absence signs of myocardial necrosis (elevation of cardiac enzymes). On the ohter hand, the European System Risk in Cardiac Operations (EuroSCORE) defines UA as anginal pain at rest that requires treatment with intravenous nitroglycerin to the surgical procedure. For purposes of this study UA was defined as acute coronary syndrome without ST elevation (ACSST) and without enzyme and/or class IV angina [6,7].
According to current recommendations on UA, taking into account the patient's risk, CABG is indicated in cases of severe injury of the left main coronary artery, three-vessel disease with impaired left ventricular function (left ventricular ejection fraction < 0.5 ); two-vessel lesion with involvement of the proximal left anterior descending artery or decreased left ventricular function or provoked ischemia. Life expectancy, associated diseases, symptom severity and amount of viable myocardium at risk are also important factors [5,8,9].
It is not well defined which the real impact of UA on the prognosis of patients undergoing CABG in this context, or whether there is an optimal time interval between the acute event and revascularization. The aim of this study is to assess the characteristics of patients presenting UA and indication of CABG and compare with patients without UA in the preoperative of CABG as well as compare the in-hospital outcomes of these patients in this context.
METHODS
Population and sample
In the period from February 1996 to July 2010 2,818 isolated CABG were performed at Hospital São Lucas. Of these, 1,016 patients ( 36.1 % ) showed UA preoperatively. UA was defined as acute coronary syndrome without ST elevation (ACSST ) and without enzyme and/ or class IV angina.
Study design
Observational historical cohort study. Data were prospectively collected and entered into the database of the postoperative unit of cardiac surgery at the Hospital São Lucas at PUCRS.
Inclusion Criteria
Patients aged over 18 years led to cardiac surgery for coronary artery bypass graft (CABG) alone.
Exclusion Criteria
We excluded from the analysis valvular surgery alone or combined with CABG, urgent or emergency CABG, acute myocardial infarction with very recent (< 30 days) ST elevation preoperatively, acute myocardial infarction without very recent surgery (< 30 days) ST segment elevation in the preoperative.
Study variables
The variables included in the analysis were:
- Presence of UA in preoperative of CABG, making comparative analysis with CABG without UA preoperatively.
- Age calculated based on the average age and also divided into groups for analysis: less than 60 and greater than or equal to 60 years.
- Gender (male/female).
- Ejection fraction (LVEF ) calculated by echocardiography, values divided for analysis in less than 40% and greater than or equal to 40%.
- Chronic kidney disease (CKD) - diagnosed by serum creatinine> 1.5 mg/dl.
- Diabetes Mellitus (DM).
- High blood pressure (HBP).
- Acute Myocardial Infarction (AMI) defined as acute coronary syndrome occurred in 30 to 90 days after surgery.
- Analysis of surgical risk by EuroSCORE.
- The need for intra-aortic balloon preoperatively (preop IAB).
- Presence of severe injury of the left main coronary artery (SL LCT) - considered as obstructive lesion greater than 50%.
- Previous use of drug: acetylsalicylic acid (ASA), beta blockers (BB), antiarrhythmics (amiodarone and propafenone), digoxin, corticosteroids, calcium channel antagonists, diuretics, statins, Heparin, angiotensin-converting enzyme inhibitors (ACEI), nitrates, oral hypoglycemic agents, insulin.
- Need for vasopressor and invasive monitoring with Swan-Ganz.
- Time of cardiopulmonary bypass (CPB), all CABG used CPB.
- Complete revascularization, considered when all vessels with a caliber greater than 1.5 mm and lesions with obstruction > 50 % were revascularized.
- Use of revascularization (all used internal thoracic artery graft).
- AMI postoperatively.
- Stroke (CVA) postoperatively.
- Atrial fibrillation (AF) postoperatively.
- Acute renal failure (ARF) in the postoperative period, considered with a 50% increase in serum creatinine.
- Increased bleeding after surgery, it is considered excessive bleeding 200 ml/hr to 3 ml/h/kg during the first two hours postoperatively, or persisting around 100 ml/h or 1.5 ml/h/kg from the third hour.
- Need for multiple blood transfusions, need for transfusion to treat increased bleeding that triggers significant anemia (hemoglobin <8.0 mg/dl) or hemodynamic changes.
- Need for reintervention.
- Length of hospital stay.
- In-hospital mortality postoperatively.
Outcome
We assessed death rates, need for vasopressor support with intra-aortic balloon and length of stay in the postoperative of CABG.
Procedures
The anesthesia techniques of cardiopulmonary bypass (CPB) and cardioplegic solution (St. Thomas No. 2) were performed according to the standardization of the Hospital São Lucas, as previously described [10]. After surgery, all patients were transferred to the ICU postoperatively in cardiac surgery, under mechanical ventilation.
Statistical Analysis
The data were plotted on a spreadsheet Microsoft Access® and assessed in SPSS version 11.0. Descriptive statistics were performed, as well as the univariate tests: Chi - square test for ordinal variables and quantitative data was used for analysis of variance or Student's t test (for unpaired variables) followed by post hoc test for Bonferroni data. Multivariate analysis was performed by logistic regression (backward conditional method). Statistical differences were considered when P<0.05.
Ethical Considerations
The research design of the study was submitted to the Research Ethics Committee of FAMED PUCRS, with protocol number 06003478.
RESULTS
In the period from February 1996 to July 2010 2,818 isolated CABG were performed, all using CPB. Of these, 1,016 patients (36.1%) showed UA preoperatively. The average age of the study population with UA preoperatively was 60.42 ± 10 years, left ventricular ejection fraction with average of 54.05±15%, CPB time of 85.54±34 minutes, average use of mammary graft in 74.1% and average rate of incomplete revascularization of 6.8%. All these characteristics were similar between the groups with UA and without UA in the preoperative of CABG.
Regarding the surgical risk analysis performed by logistic EuroSCORE, the average of surgical risk of patients with UA in preoperative of CABG was 5.19 compared with 3.30 for patients without UA in preoperative P=0.012. The mean hospital stay was 10.96±9.74 days, and waiting times for CABG in total was on the average 9.18 days, with longer interval for patients with UA (11.7 days) compared with no UA (7.8 days) (OR 1.30, 95% CI 1.29 to 6.41, P=0.003). Postoperative time was similar between groups, in total the average was 10.5 days, in patients with UA 10.99 days and without UA 10.27 days (OR:0.36, 95% CI 0.006 - 1.42, P=0.048). The longer length of stay in patients with UA preoperatively (10.96 days versus 10.27 days) was at the expense of longer waiting times for CABG.
The preoperative and postoperative characteristics with univariate analysis are described in Tables 1 and 2, respectively. Table 3 describes the characteristics and clinical outcomes in postoperative of CABG alone in patients with UA preoperatively, with statistical significance.

Univariate analysis showed that females were more prevalent among patients with UA in preoperative of CABG, in addition, these patients used more IAB preoperatively and medications such as aspirin, angiotensin-converting enzyme angiotensin nitrate, heparin and beta-blockers. But the use of diuretics were lower in this group of patients (Table 1).
On the evolution postoperatively, there was no difference in outcomes such as myocardial infarction, stroke or acute renal failure and need for vasopressor among patients with UA preoperatively and those who did not. Even with the highest rate of use of aspirin and heparin in patients with UA in preoperative of CABG, there was no difference between groups in relation to bleeding: 12 % in the group with UA and 10.7 % in the group without UA (OR: 1.14, 95% CI 0.89 to 1.45, P=0.29), need for reintervention: 4.1% in the group with UA and 5.3 % in the group without UA (OR: 0.77 95% CI 0.53 to 1.12, P=0.17), and need for multiple transfusions tended to be higher in patients with UA (OR: 1.22, 95% CI 1.02 to 1.45, P=0.027), not confirmed in the multivariate analysis. The CPB time was similar between the groups with values > 90 minutes in 37.9 % in the group with UA compared to 40.7% without UA (OR: 0.89, 95% CI 0.75 to 1.04; P=0.98), use of Swan-Ganz was higher in patients with UA preoperatively (Table 2).
Multivariate analysis showed that patients with UA in the preoperative of CABG were mostly female, used more medications such as aspirin, beta-blockers, heparin (anticoagulation), nitrate and needed less diuretics. More patients received monitoring with Swan-Ganz and support with intra-aortic balloon (Table 3). The overall rate of death in the study population was 5.4%, and the patients with UA had a lower death rate, 4.1% compared to the UA without preoperative rate of 6.1%, data with statistical significance (OR: 0.64, 95% CI 0.443 to 0.925, P=0.018), with a protective effect of 36% of deaths in patients with UA in preoperative of CABG. The hospitalization was higher in patients with UA requiring 10.96 days compared to 10.27 days for patients without UA (OR: 1.009, 95% CI 1.001 to 1.018, P=0.030), this difference was due to higher waiting time for CABG for patients with UA (Table 3).
Given the long period examined, from 1996 to 2010, and considering the changes in the management of acute coronary syndromes during this period, including UA, as the benefit of beta-blockers, statins, angiotensin-converting enzyme inhibitors and maintenance of acetylsalicylic acid for patients with UA undergoing CABG, we performed an analysis comparing two periods, as shown in Figures 1 and 2. Through this analysis, we conclude that patients with UA treated from 1996 to 2003 received more beta blockers, angiotensin-converting enzyme inhibitors, aspirin, Heparin and nitrate, less use of diuretics, and the death rate (3% versus 5.3%) was significantly lower in this group compared to those without UA, P=0.028. On the other hand, patients treated from 2004 to 2010 with UA received more aspirin, statins, Heparin and nitrate, and less use of diuretic, but no difference in the treatment of beta-blockers, angiotensin-converting enzyme inhibitors, showing similar mortality rate (6.4 % versus 7.7%) and higher in comparison with the previous period, perhaps for the greater complexity of patients undergoing CABG with the evolution of time (older, with more comorbidities).
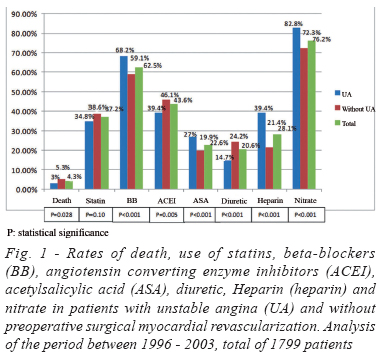
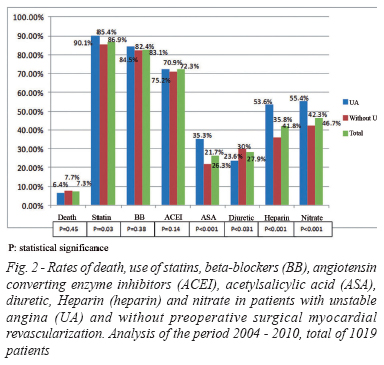
DISCUSSION
The risk scores that estimate the risk of operative mortality and morbidity of heart surgery in adults based on demographic and clinical variables, such as the European System Risk in Cardiac Operations (EuroSCORE) and the Society of Thoracic Surgeons (STS), consider the UA as additional risk to patients undergoing CABG, deserving score in their scores, but when alone does not alter the risk, so when presenting this feature as an isolated risk factor it remains as low risk. On the other hand, score developed at the Hospital São Lucas/PUCRS found no increased risk of operative mortality and in-hospital postoperative of CABG in patients with signs of UA, not receiving score [11].
The use of Swan-Ganz catheter was significantly higher in patients with UA in preoperative of CABG. Most invasive monitoring was not associated with higher rates of cardiogenic shock, postoperative infarction or sepsis, since the use of diuretics was significantly lower in this group of patients (OR 0.57), possibly due to the use of this tool. The increased use of Swan-Ganz could be related as a marker of severity for this group of patients. Nevertheless, obtaining specific hemodynamic parameters, contributed to lower outcomes postoperatively.
Monitoring of fluid replacement is a fundamental aspect to be avoided complications caused by overload or lack of volume in the intravascular space. The use of invasive hemodynamic monitoring in the postoperative period of cardiac surgery in patients with hemodynamic instability is useful in guiding the control of plasma volume, use of vasoactive drugs, requiring circulatory assistance. Another fact to be noted is that there is no reliable correlation between central venous pressure (CVP) and pulmonary capillary pressure (PCP), and the control of blood volume according to CVP is not safe [12]. However, studies show that these values are somewhat inaccurate with respecti to assessment of blood volume, especially in patients on mechanical ventilation due to changes in systolic volume induced by preload also which depend on contractility and afterload, which are not assessed by these parameters.
Other methods such as echocardiography and transesophageal Doppler with measurements of aortic flow and end-diastolic volume are interesting options with drawback of relying on experienced operator and can not be used continuously for a long period of time. Another sensitive indicator of response to blood volume in mechanically ventilated patients is the change in systolic blood pressure and its ddown, with good correlation with negative deflection and fluid infusion and concomitant improvement in cardiac output and increase in atrial filling pressures [13].
The need for intra-aortic balloon was also significantly higher in the group with UA, which may represent the most severity of these when compared to the group without acute event in the preoperative of CABG alone. The indication of intra-aortic balloon in our service is limited to low ejection fraction (< 30 %), cardiogenic shock, severe injury to the left main coronary (> 50 %) and intractable angina, the latter being the main reason for using this study. Even needing this support preoperatively, death rates were lower.
The benefits of intra-aortic balloon were studied by Santarpino et al. [14], which assessed the use of intra-aortic balloon in the preoperative period of CABG of high risk (EuroSCORE>12) and found similar outcomes to low risk CABG (EuroSOCORE<5) without the device, with low rates of associated complications, advocating a more liberal use of intra-aortic balloon, especially in high-risk CABG. Macruz et al. [15] retrospectively assessed the early (intraoperative) and late (postoperative) use of intra-aortic balloon. 130 patients were studied at Texas Heart Institute. Data from this study suggest that the intra-aortic balloon is effective as a method of circulatory support in patients with low cardiac output after cardiopulmonary bypass, with a tendency to better results when used early; it was observed influence of age and CPB time, suggesting that more efficient methods of circulatory support should be used in elderly patients and in patients with low cardiac output syndrome after cardiac surgery with CPB times greater than 120 minutes.
Females had an odds ratio of 1.4 for UA in the preoperative of CABG. It is believed that the behavior of coronary heart disease is different between genders. Campbell et al. [16] compared the histological structure of the myocardium and coronary microvasculature of men and women and found differences in the arterioles wall and the ratio of the cardiac myocyte and body surface area that can justify the presence of ischemic symptoms in more frequent in females, although not always correlating with more severe coronary artery disease than in males. Hasdai et al. [17] assessed the outcome of 10,000 patients after ACS in relation to gender and observed in this record that females had older age, no association with increased mortality, lower use of antiplatelet agents (such as glycoprotein IIbIIIa) and less use of less beta blockers, antiplatelet and statin, and more diuretics and digoxin at discharge [17]. Although studies suggest female as a risk indicator for CABG and being part of the scores [18], others did not confirm this risk in the context of ACS [19-21], as well as the present study.
The length of stay was longer in patients with UA in preopearative of CABG, at the expense of longer wait times for CABG, and not due to prolonged ventilation, need for vasopressors, sepsis and delirium. There are no clear recommendations on which the ideal timeout for conducting CABG after UA. The advantages of early CABG for the treatment of UA include limiting the area of ischemia and left ventricular remodeling, however, it can lead to reperfusion injury and further damage to the myocardium, thereby increasing the systemic inflammatory response [22].
The optimal time interval between the ACS and CABG is still controversial. The PL-ACS registry included 2028 patients with acute coronary syndrome (ACS), in which 55% had UA and indication for CABG, 60% underwent surgery at different time intervals. They concluded that after 12 months of waiting for CABG the risk of death rises in twice and CABG performed during the first month after ACS is associated with increased prevalence of mortality, considering the ideal interval to wait between 1-3 months [22]. This result is different compared to the found in this study, in which the average waiting time was 11.7 days; unrelated to increased mortality, probably due to not including patients with acute transmural myocardial infarction.
Weiss et al. [23], in a study with analysis of records of more than 40,000 patients with ACS, including transmural infarction, observed that patients undergoing CABG in 2 days of hospitalization had higher mortality rates than those who were taken to surgery with waiting time of three or more days of hospitalization in non-emergency cases, suggesting that the optimal time interval between CABG after ACS should be greater than or equal to 3 days. Costa et al. [24] found similar conclusion after comparing outcomes between two groups of patients waiting for less than 30 days and more than 30 days undergoing CABG: overall mortality was 5.9 % (1 death in 15 patients in Group I and 1 death in 19 patients in group II - P=1.0); they argue that CABG can be performed safely, especially after the first 72 hours of the ischemic event.
The UA in preoperative of CABG alone, different from the expected, showed a lower rate of in-hospital mortality in this study, compared with the group without UA, even with higher surgical risk by assessing the logistic EuroSCORE in this group (5.19 compared with 3.30 of the patients without preoperative UA P=0.012). Possibly by the fact that this sample of patients with signs of UA has received optimized medical treatment, compared with the group with no UA, such as greater use of acetylsalicylic acid, beta-blockers, full-dose heparin and nitrate (OR 1.62; 1.19, 2.01, 1.63 , respectively) compared with those who did not have the acute event. But the use of diuretics was significantly lower in this group of patients (OR 0.57). This fact reveals the great importance in medical management for all groups.
The average mortality rate was 5.4 %. Mortality in patients with preoperative UA undergoing isolated CABG was 4.1 %, versus 6.1% in the group without acute event in this study. The death rate is similar to that presented by other authors as Kaul et al. [18] (5.9%), Hochman et al. [25] (4.3%), Howard et al. [26] (4.3%) and Jatene et al. [21] (4.1%), in the context of ACS. However, when we compared the mean death for periods, we observe that there is a significant difference when assessing the period 1996-2003 (4.3%) and 2004-2010 (7.3%) P=0.001, despite advances in treatment and management of ischemic patients undergoing CABG, with greater use of statins, beta-blockers, angiotensin-converting enzyme inhibitors and acetylsalicylic acid, which can be explained by the increased complexity of patients undergoing CABG, with more comorbidities, age and more complex pathologies.
When assessing patients with UA undergoing isolated CABG in the period 1996-2003, we found that this group of patients showed a lower mortality rate compared to those without UA and that there was no significant difference in death rate between the patients with and without UA in the period 2004-2010. In this period, the optimal medical treatment did not appear to reduce mortality, perhaps because both groups of this period present more uniform clinical treatment and management, particularly regarding the use of beta-blockers and angiotensin converting enzyme angiotensin.
Limitations
Although the data collection was performed prospectively, the data analysis was performed retrospectively, and data were not collected for the specific purpose of this analysis. Thus, some important issues were not raised as a risk assessment according to the scores of acute coronary syndrome as TIMI score or GRACE, which would enable a more detailed assessment regarding the severity of patients preoperatively. Another limitation is the monitoring of the patient, performed until hospital discharge of which we have no data on outcomes after this time.
CONCLUSION
The present study showed that patients with UA undergoing isolated CABG showed no increased mortality. Rather, possibly because they have received better medical treatment, and invasive monitoring and hemodynamic support more frequently, patients with UA had lower death rates than those considered stable.
REFERENCES
1. Movahed MR, John J, Hashemzadeh M, Hashemzadeh M. Mortality trends for non-ST-segment elevation myocardial infarction (NSTEMI) in the United States from 1988 to 2004. Clin Cardiol. 2011;34(11):689-92. [MedLine]
2. Feyter PJ, Serruys PW, Unger F, Beyar R, Valk V, Milo S, et al. Bypass surgery versus stenting for the treatment of multivessel disease in patients with unstable angina compared with stable angina. Circulation. 2002;105(20):2367-72. [MedLine]
3. Luksienè D, Milvidaitè I, Slapikas R, Jarusevicius G, Siudikas A, Venclovienè J, et al. The impact of myocardial revascularization after acute coronary syndromes on one-year cardiovascular mortality. Medicina (Kaunas). 2011;47(6):305-12. [MedLine]
4. Weber MM. Surgical management of unstable angina and symptomatic coronary artery disease. J Cardiovasc Nurs. 2000;15(1):27-42. [MedLine]
5. Sociedade Brasileira de Cardiologia. Guidelines for Unstable Angina and Non-ST-Segment Elevation Myocardial Infarction of the Brazilian Society of Cardiology (II Edition, 2007). Arq Bras Cardiol. 2007;89(4):e89-131. [MedLine]
6. Bonow RO, Zipes DP, Libby P. Braunwald: tratado de doenças cardiovasculares. 8ª ed. Rio de Janeiro:Elsevier; 2009. p.1319.
7. Naschef SA, Roques F, Michel P, Gauducheau E, Lemeshow S, Salamon R. European system for cardiac operative risk evaluation (EuroSCORE). Eur J Cardiothorac Surg. 1999;16(1):9-13. [MedLine]
8. Hillis LD, Smith PK, Anderson JL, Bittl JA, Bridges CR, Byrne JG, et al. 2011 ACCF/AHA Guideline for Coronary Artery Bypass Graft Surgery: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2011;124(23):e652-735. [MedLine]
9. Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS); European Association for Percutaneous Cardiovascular Interventions (EAPCI), Wijns W, Kolh P, Danchin N, Di Mario C, Falk V, Folliguet T, et al. Guidelines on myocardial revascularization. Eur Heart J. 2010;31(20):2501-55. [MedLine]
10. Guaragna JCVC. Cirurgia cardíaca e hipertensão arterial no pós-operatório imediato: fatores pré e transoperatórios [Dissertação]. Porto Alegre: Pontíficia Universidade Católica do Rio Grande do Sul; 1999.
11. Cadore MP, Guaragna JCVC, Anacker JFA, Albuquerque LC, Bodanese LC, Piccoli JCE, et al. Proposição de um escore de risco cirúrgico em pacientes submetidos à cirurgia de revascularização miocárdica. Rev Bras Cir Cardiovasc. 2010;25(4):447-56. [MedLine] View article
12. Dragosavac D, Araújo S, Carieli MCM, Terzi RGG, Dragosavac S, Vieira RW. Monitorização hemodinâmica invasiva no pós-operatório de cirurgia cardíaca. Arq Bras Cardiol. 1999;73(2):129-38. [MedLine]
13. Carlos RV, Bittar CS, Lopes MR, Auler Júnior JOC. Variação da pressão sistólica como método diagnóstico da hipovolemia durante anestesia para cirurgia cardíaca. Rev Bras Anestesiol. 2005;55(1):3-18. [MedLine]
14. Santarpino G, Onorati F, Rubino AS, Abdalla K, Caroleo S, Santangelo E, et al. Preoperative intraaortic ballon pumping impoves outcomes for high-risk patients in routine coronary artery bypass graft surgery. Ann Thorac Surg. 2009;87(2):481-8. [MedLine]
15. Macruz HMS, Frazier OH, Cooley DA. Análise dos resultados do uso precoce e tardio da assistência circulatória com balão intra-aórtico (BIA) em pacientes submetidos a correção de cardiopatias com auxílio de circulação extracorpórea. Rev Bras Cir Cardiovasc. 1995;10(4):190-7. View article
16. Campbell DJ, Somaratne JB, Jenkins AJ, Prior DL, Yii M, Kenny JF, et al. Differences in myocardial structure and coronary microvasculature between men and women with coronary artery disease. Hypertension. 2011;57(2):168-92.
17. Hasdai D, Porter A, Rosengren A, Behar S, Boyko V, Battler A. Effect of gender on outcomes of acute coronary syndromes. Am J Cardiol. 2003;91(12):1466-9.
18. Kaul TK, Fields BL, Riggins SL, Dacumos GC, Wyatt DA, Jones CR. Coronary artery bypass grafting within 30 days of an acute myocardial infarction. Ann Thorac Surg. 1995;59(5):1169-76. [MedLine]
19. Naunheim KS, Kesler KA, Kanter KR, Fiore AC, McBride LR, Pennington DG, et al. Coronary artery bypass for recent infarction. Predictors of mortality. Circulation. 1988;78(3 Pt 2):I122-8. [MedLine]
20. Applebaum R, House R, Rademaker A, Garibaldi A, Davis Z, Guillory J, et al. Coronary artery bypass grafting within thirty days of acute myocardial infarction. Early and late results in 406 patients. J Thorac Cardiovasc Surg. 1991;102(5):745-52. [MedLine]
21. Jatene FB, Nicolau JC, Hueb AC, Atik FA, Barafiole LM, Murta CB, et al. Fatores prognósticos da revascularização na fase aguda do infarto agudo do miocárdio. Rev Bras Cir Cardiovasc. 2001;16(3):195-202. View article
22. Zembala M, Trzeciak P, Gasior M, Gierlotka M, Hudzik B, Taistra M, et al. Optimal timing for surgical revascularization in survivors of acute coronary syndromes elegible for elective coronary artery bypass graft surgery. Int J Cardiol. 2011;153(2):173-8. [MedLine]
23. Weiss ES, Chang DD, Joyce DL, Nwakanma LU, Yuh DD. Optimal timing of coronary artery bypass after acute myocardial infarction: a review of California discharge data. J Thorac Cardiovas Surg. 2008;135(3):503-11.
24. Costa WA, Santos JMV, Sampaio DT, Lobo Jr. NC, Figueroa CCS. Cirurgia de revascularização do miocárdio no paciente infartado: quando operar? Rev Bras Cir Cardiovasc. 1999;14(1):19-26. View article
25. Hochman JS, Boland J, Sleeper LA, Porway M, Brinker J, Col J, et al. Current spectrum of cardiogenic shock and effect of early revascularization on mortality. Results of an International Registry. SHOCK Registry Investigators. Circulation. 1995;91(3):873-81. [MedLine]
26. Howard M, Irarrazaval MJ, Corbalán R, Morán S, Zalaquett R, Maturana G, et al. Surgical myocardial revascularization during the 1st 15 days of evolution of acute myocardial infarction. Rev Med Chil. 1996;124(1):37-44. [MedLine]
Authors' roles & responsibilities
CPS: Main author
JCG: Coauthor
RSC: Coauthor
JP: Coauthor
LCA: Coauthor
MAG: Coauthor
JBP: Coauthor
LCB: Coauthor
Article receive on Monday, June 25, 2012
 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license