Fábio Antonio GaiottoI; Caio B. ViannaI; Fábio F BusnardoII; José R. PargaI; Luis Alberto de Oliveira DallanI; Luis A. M. CesarI; Noedir A. G. StolfI; Fabio B. JateneI
DOI: 10.5935/1678-9741.20130050
DLFCA: Descending branch of the lateral femoral circumflex artery
CABG: Coronary artery bypass grafting
LITA: Left internal thoracic artery
SV: Saphenous vein
FAG: Study design, surgical procedures and manuscript writing
CBV: Patients clinical follow up
FFB: Orientation in the preparation of the arterial graft
JRP: Angiotomography evaluation
LAOD: Angiotomography evaluation
LAMC: Patients clinical follow up
NAGS: Orientation for study design and final manuscript approval
FBJ: Orientation for study design and final manuscript approval
INTRODUCTION
It is well known that arterial grafts provide better patency rates for coronary artery bypass grafting (CABG) than saphenous veins (SV), in both the short and long-term [1-5]. Therefore, alternative arterial grafts have been continuously researched. The most commonly used arterial grafts are the left and right internal thoracic [1,2,5-8] and radial arteries [9,10]. The inferior epigastric and gastroepiploic arteries are less frequently used [1,5,7,10]. Other arterial conduits, such as the splenic, subscapular, inferior mesenteric, ulnar, and intercostal arteries have rarely been used [5].
In plastic and reconstructive surgeries, the lateral femoral circumflex artery is often used to supply composite skin and muscle tissues, such as an anterolateral thigh flap [11,12]. The descending branch of the lateral femoral circumflex artery (DLFCA) has attractive characteristics that qualify it as an option for arterial grafting in surgical myocardial revascularization.
The first case in which the DLFCA was used as a graft in CABG was performed in Japan and was reported by Tatsumi et al. [13], in 1996, when the DLFCA was used as a free graft from the aorta to the right coronary artery. Two years later, Schamún et al. [14] reported promising results using the DLFCA as a coronary graft in 35 patients, with most cases employing early evaluation via angiographic studies after surgery. In these series, the left anterior descending coronary artery was revascularized by the left internal thoracic artery (LITA) in all patients, and the DLFCA was used as a composite graft with the LITA (end-to-side with a Y shape) in most cases. In a few patients, the DLFCA was used as an extension of the right internal thoracic artery (end-to-end anastomosis to revascularize the distal portion of the right coronary artery) or as a free aorta-coronary graft. In 2003, Fabbrocini et al. [15] published the most relevant study on this topic, reporting excellent clinical results and patency rates using the DLFCA as a composite graft with the LITA, in 147 patients, in the short and mid-term.
We evaluated the early outcomes, technical results, patency rates and lumen diameter adaptation of the DLFCA when used as a composite graft with the LITA or SV, by multidetector computed angiotomography.
METHODS
Patient Selection
During a period of one year and eight months, a single surgical team selected 32 patients as candidates for CABG using the DLFCA as a composite graft. All patients had stable three-vessel coronary artery disease with normal or nearly normal left ventricular function. Inclusion criteria were absence of peripheral arteriopathy and critical stenosis or oclusion of coronary branches. In patients with indication for doble mammary, DLFCA was used as a complementation. All patients with suspected or established lower limb arterial disease, renal insufficiency (defined by a creatinine serum level >2 mg/dL or the patient being on dialysis), and previous cardiac operation were not included. The institutional committee on human research approved the study and all patients gave informed consent regarding the study protocol.
Study Protocol
After median sternotomy, the LITA and SV were simultaneously dissected, and the DLFCA was dissected soon thereafter. All coronary arteries were revascularized on cardiopulmonary bypass with normothermic perfusion and warm myocardial protection. Composite conduits of the DLFCA with the LITA were prepared (end-to-side, in a Y shape) before initiating extracorporeal circulation and were covered with gauze in physiologic saline solution and papaverine. After initiating extracorporeal circulation, the left anterior descending coronary artery was revascularized by the LITA, and the scheduled revascularizations with SV were accomplished. Other arterial grafts were used infrequently: radial artery in one and right internal thoracic artery in 4 cases. Composite grafts of the DLFCA with SV were constructed at the end of the operation while the patients were on cardiopulmonary bypass. The proximal anastomosis of the DLFCA with SV was performed near the origin of the vein in the aorta. On-pump, all composite DLFCA grafts were anastomosed onto a previously selected coronary artery with an acceptable lumen diameter and atherosclerotic obstruction of 70% or more. All anastomoses were performed using a continuous 8-0 polypropylene suture.
The DLFCA was approached through an incision 15 to 20 cm in length on the anterolateral face of the thigh in its proximal and middle third along the lateral margin of the rectus femoris muscle (Figure 1A). The incision corresponded to an imaginary straight line between the anterosuperior iliac spine and the lateral border of the patella. The fascia lata of the quadriceps was incised longitudinally. Generally, the DLFCA arises from the lateral femoral circumflex artery soon after its origin from the deep femoral artery, infrequently arising separately. The DLFCA courses downward through the intermuscular space between the rectus femoris and the vastus lateralis muscles (Figure 1B) near the femur, ending in the vastus muscle near the knee joint. Harvesting was performed carefully with minimal handling using very low-voltage electrocautery.
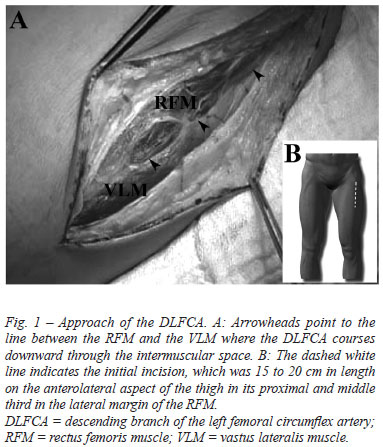
The dissection was started from the beginning of the DLFCA toward the end in a non-skeletonized fashion (Figure 2A). The two large veins that usually accompany the artery were left undisturbed, the femoral nerve branches were not involved, and all collateral branches were ligated for complete dissection. The distal portion of the DLFCA harvesting was cut, and a solution with papaverine was injected through it. The proximal portion was then cut, the harvesting was removed, and composite conduits of the DLFCA with the LITA or SV were prepared (Figure 2B).
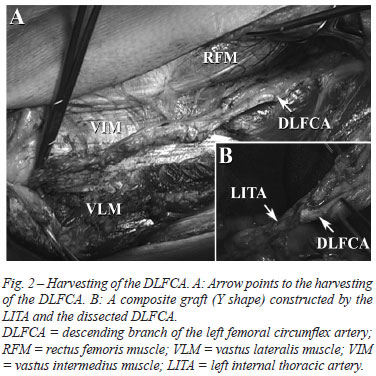
However, prior to construction, to prevent vasospasm, the dissected DLFCA was subjected to alternating submersions in two 0.9% saline solutions at 36ºC: one with papaverine (1:20 dilution); another with nitroglycerin (10 mg) and diltiazem (50 mg) diluted in a physiologic solution (500 ml). Additionally, to avoid arterial vasospasm, the patients were continuously supplied with intravenous nitroglycerin throughout the operative period, and oral diltiazem was administered soon after endotracheal extubation and maintained for three months.
Computed Angiotomographies
Computed tomography images were acquired with a multidetector system (Aquilion® 64, Toshiba Medical Systems, Japan) using previously described parameters [16] approximately 1 week after CABG, and were repeteaded 3 months later. Briefly, all patients with contraindication to contrast computed tomography were excluded and scanning was planned based on scout images to cover the entire extension of the chest from the LITA to the bottom of the heart. Unless contraindicated, patients with a heart rate above 60 bpm received intravenous metoprolol at a dose of up to 15 mg to reach between 50 and 60 bpm. Patients with a contraindication to beta-blockers were given diltiazem intravenously (0.25 mg/kg; if necessary, a second dose was given to reach the expected heart rate). Sublingual nitrates were administered just before scanning in all patients in whom it was not contraindicated (systolic blood pressure <110 mmHg, intake of phosphodiesterase inhibitors, known intolerance) in order to prevent vasospasm and increase vessel size.
Scan parameters were adjusted by body mass index and a prospective acquisition electrocardiogram gating (55% to 85% of the RR), with heart rate-adjusted gantry rotation of 350-500 msec, pitch and tube currents of 240-450 mA, 120-kV tube voltage, 0,5 mm slice thickness to ensure a 12-15 mSv radiation dose. The patients were then injected with 120 ml of nonionic iodinated contrast (Iopamiron® 370, Bayer Schering Pharma AG, Berlin, Germany) at a rate of 5 ml/s through the antecubital vein, followed by 30 ml of saline solution. The arrival of the contrast was monitored through the acquisition of axial images at the level of the descending aorta 10 s after infusion of contrast (SureStart, Toshiba Medical Systems, Japan). Acquisition of the contrasted set began automatically when the threshold of 180 Hounsfield units was achieved in the region of interest (descending aorta). For image reconstruction a retrospective analysis of the electrocardiographic tracing was employed, searching for the best image during the middle diastole, generally approximately 700 ms before the next R wave or 75% of the RR. From this set, the entire block was reprocessed with a thickness of 0.3 mm and analyzed in a workstation (Vitrea®, Vital Images, USA).
The angiotomographies purposes were to verify patency rates, to identify technical problems (kinks and anastomotic stenoses), and to measure the lumen diameters of the DLFCA and LITA grafts at three points: 3 cm after the proximal anastomosis, in the middle of the graft, and 3 cm before the distal anastomosis. All native vessels and grafts were evaluated with transverse, coronal, sagittal, oblique and reformatted curved planes to visually identify proximal and distal anastomosis. Anastomotic stenoses were considered if the size of the proximal vessel was greater than the anastomotic site, with a 50% cutoff considered significant. Special attention was paid to bypass grafts as they were the most important data in the study. All measurements were manually traced three times at the designated portions of the vessel by a blinded reader to the design of the study (>7 year coronary experience in computed tomography). The mean value of the automatic software results were used as the reference value for comparison.
Statistical Analysis
Quantitative data were expressed as mean±SD, and discontinuous values were presented as percentages. Quantitative values were compared using a two-sided paired Student's t test or Wilcoxon signed rank test for non-Gaussian distributions. All data analyses were performed using the Statistical Package for the Social Sciences (version 17.0, SPSS Inc., Chicago, IL, USA). Differences at P<0.05 were considered to be statistically significant.
RESULTS
After dissecting the DLFCA in the operating room, out of the 32 selected patients, the artery was considered appropriate for use in 26 (81%); 22 of whom were male (85%), aged 62±7 years (range, 44 to 73 years-old). All patients had stable triple-vessel coronary artery disease. Hypertension was present in 92% of the patients and diabetes mellitus in 26%. In six (19%) patients, four of them being male, aged 69±6 years (range, 60 to 75 years), the DLFCA was considered inappropriate for use as a graft because of the following: it was not found in one case; one had severe atherosclerosis; two were very short; and two were short and thin. The mean length of the grafts obtained after dissection was 9.9±4.0 cm (range, 4 to 19 cm) in the 26 patients with an appropriate DLFCA. Among the six cases with inappropriate grafts, except for the cases with a missing DLFCA and severe atherosclerosis, the mean length of the grafts obtained was 5.2±2.0 cm (range, 4 to 8 cm). Thus, the standardized surgical protocols were applied in 26 of the 32 selected patients. The mean cardiopulmonary bypass time was 85 ± 27 minutes (range, 50 to 150 minutes), and the aortic cross clamp time was 58±21 minutes (range, 34 to 110 minutes).
All of the intraoperative and immediate postoperative periods were uncomplicated. On average, 3.7±0.8 (range, 3 to 5) coronary arteries were grafted for each patient. The composite DLFCA grafts were constructed with the LITA (Y shape) in 15 cases and with SV in 11 cases. The coronary arteries revascularized by the DLFCA grafts were as follows: diagonal branch of the left anterior descending artery in 13 patients, obtuse marginal branch of the circumflex artery in eight, intermediate left main coronary artery in three, right coronary artery in one, and posterolateral branch of the right coronary artery in one patient.
No clinical complications related to the LITA or DLFCA grafts were observed during the hospital stay, after surgery, or during the 3-month follow-up period after discharge. No ischemic event, re-exploration for bleeding, sternal infection, or mediastinitis was observed in any patient.
The first computed angiotomography was performed in-hospital in all patients, 7±1 days (range, 5 to 11 days) after the operation, and it was repeated 90±5 days (range, 83 to 103 days) after surgery. Despite previously signing the consent form, one patient refused to undergo the tomographies after surgery. In another patient, the images of the first tomography were poor and the exam was not repeated on the 90th day after surgery. One angiotomography performed 3 months after surgery was of sufficiently good quality to measure the lumen of the DLFCA graft, but not of the LITA.
As a result, the inner diameters of the DLFCA graft were measured in 24 cases, and those of the LITA graft were determined in 23 patients. The patency rates of the DLFCA grafts were 23/24 patients (96%) on the 7th day and 22/24 patients (92%) on the 90th day after surgery. The patency rates of the LITA grafts were 100% in both evaluations. Signs of vasospasm and technical complications, such as kinks in the grafts or anastomotic stenosis, were not observed in any case. The results regarding the inner diameters of the grafts are presented in Table 1. Comparison of the lumen values between the 7th and 90th day after surgery showed positive adaption both of the DLFCA and LITA grafts. The lumen of the DLFCA grafts increased significantly in the middle and distal portions, and the lumen of the LITA grafts increased significantly in the middle portion (Table 1).
DISCUSSION
Although the DLFCA is a recognized alternative arterial graft for CABG, especially as a composite conduct, little information is available regarding the outcomes, patency, vasospasm and the occurrence of technical complications, such as kinks or anastomotic stenosis. This study sought to evaluate these data in the short-term.
Some authors recommend femoral arteriography before every CABG [13,17] in which the DLFCA is a planned graft because this vessel can present anatomical variations, such as short length, narrow lumen diameter or anomalous origins, that make it difficult to dissect and use [18]. Another reason to perform preoperative femoral arteriography is to verify the presence of atherosclerosis. Despite this, Schamún et al. [14], in 1998, did not perform femoral angiographies before CABG in the 35 patients addressed in their report, but only carried out careful clinical preoperative evaluations to rule out lower limb arterial disease. Similarly, in the series of Fabbrocini et al. [15] (147 patients undergoing CABG using DLFCA grafts) the authors performed preoperative continuous-wave Doppler to investigate arteriosclerosis in the iliac and femoral arteries. When significant arterial disease was discovered, femoral arteriography was performed to assess the DLFCA. Our study design included only a careful clinical preoperative evaluation, and patients with suspected or proven lower limb arterial disease were excluded.
However, our results showed that in six of the 32 selected patients the DLFCA was inappropriate for use as a graft since one presented severe atherosclerosis, one was not found, and four had very short length and/or narrow lumen diameter. These six cases were excluded of the study in the operating room. We deemed a 19% exclusion rate to be too high. In fact, we changed our initial opinion and concluded that some preoperative femoral arteriographic evaluation is mandatory. Preoperative continuous-wave Doppler, as performed by Fabbrocini et al. [15], can be used to investigate arteriosclerosis, but not anatomical variations of the DLFCA, which are the most common problem. Femoral angiotomography is a non-invasive type of arteriographic evaluation that can identify anatomical variations and atherosclerosis, thus avoiding dissection of the DLFCA in some patients and the aesthetic consequences of a thigh incision.
No clinical complications related to the LITA or DLFCA grafts were observed during the hospital stay, after surgery, or during the 3-month follow-up period after discharge, and vasospasm was not noted. No ischemic event, re-exploration for bleeding, sternal infection, or mediastinitis was observed in any patient. No significant complications in lower limbs occurred after dissection of the DLFCA in any of the patients. The surgical wound healed rapidly with no infection or compartment syndrome. Transient dysesthesia of the thigh was observed in only one patient. All patients were discharged. These results indicated very satisfactory early outcomes after surgery.
The in-hospital angiotomographic evaluation of the patients (on the 7th day after surgery) did not show vasospasms, kinks or technical complications in the anastomoses, and showed very good patency rate both of the LITA and the DLFCA grafts (100% and 96%, respectively). The only patient with an early occlusion of the DLFCA graft had no clinical complications. Furthermore, the angiotomographic evaluation performed on the 90th day of follow-up did not show any mechanical problems and revealed good patency rates (100% for the LITA graft and 92% for the DLFCA graft). Vasospasms, kinks and anastomotic stenoses were not observed on the 90th day after surgery either. Although these were early results, they were encouraging with respect to using the DLFCA as a graft in CABG.
To our knowledge, this was the first study that evaluated early adaptation of lumen diameters of the DLFCA and compared it with the LITA when used as grafts in CABG. As expected, on the 90th day following surgery, the LITA showed a significant increase in the middle portion of the lumen. The DLFCA grafts also presented positive adaptation, which was statistically significant in the middle and distal portions. These were also positive, encouraging results. We should not assert that we evaluated remodeling of the grafts because this is a broader concept involving histological and structural changes. However, we assessed variation of inner diameters, which is an important part of the remodeling.
Harvesting the DLFCA is usually quick and safe. Similar to all surgical techniques it depends on previous training. Dissection of the DLFCA should be performed by two surgeons because the quadriceps muscle is bulky and its exposure can be difficult. In our experience, initial dissections on cadavers require 30 to 40 minutes. During the surgery, harvesting is usually carried out quickly, in approximately 15 minutes. The fascia lata of the quadriceps should be closed after dissection to prevent herniation of the rectus femoris and vastus lateralis muscles. As observed in our results, a sensory deficit due to manipulation of the quadriceps nerve branches is very uncommon and usually transient [14,15]. To our knowledge, there are no reports of serious functional deficits of lower limbs or compartment syndrome arising from dissection of the DLFCA. The wound usually heals rapidly, and local infection is very uncommon. The aesthetic result is usually good because the lateral face of the thigh has no folds, and the healing line is thin and without retractions.
Computed angiotomography in the evaluation of bypass grafts still has limitations such as high attenuation artifacts attributable to beam-hardening and partial volume-averaging effects. Surgical clips and respiratory artifacts are other sources of errors. However, the new generation of scanners is getting faster and more efficient, thereby avoiding many of these drawbacks. Reports have shown the strength of the method in the evaluation of grafts after CAGB [19-24]. Obviously, conventional angiography remains the gold standard method to assess graft patency and stenoses, but serious complications such as stroke, aortic dissection, coronary dissection, and other vascular complications can occur. Nowadays, the authors are convinced that computed angiotomography is the most appropriate method to assess graft patency after CABG in study protocol involving patients [24].
The present study has evident limitations. The study is underpowered due to the small sample size and the short-term follow-up evaluation of graft patency. In fact, the authors did not intend to describe a new possibility of arterial graft, since the surgical technique and the patency rates have been previously evaluated [14,15]. The intention of the authors was to emphasize this viable alternative of using arterial graft in CABG and to evaluate the adaptation of lumen diameter. After the relevant publication of Fabbrocini et al. [15], in 2003, additional studies evaluating DLFCA grafts were no longer performed.
In conclusion, the original design of our study did not include preoperative femoral arteriography. However, in the operating room, we observed a high rate of patients (19%) in whom the graft was considered inappropriate for use due to anatomical variations, such as short length, narrow lumen diameters, and silent atherosclerosis. These results led us to change our initial opinion, and we concluded that a preoperative femoral arteriographic or ultrassonographic evaluation was mandatory for all scheduled patients, as previously recommended by some authors [13,17]. Dissection of the DLFCA graft was usually simple, quick, free from local complications, and had good aesthetic results. We used the DLFCA in composite grafts with the LITA or SV, and we observed very promising early results with regard to outcomes, technical results, patency rates and lumen diameter adaptation of this graft. The DLFCA should be considered as representative of a good alternative arterial graft in CABG.
REFERENCES
1. Fitzgibbon GM, Kafka HP, Leach AJ, Keon WL, Hooper GD, Burton JR. Coronary bypass graft fate and patient outcome: angiographic follow-up of 5,065 grafts related to survival and reoperation in 1,388 patients during 25 years. J Am Coll Cardiol. 1996;28(3):616-26. [MedLine]
2. Tatoulis J, Buxton BF, Fuller JA, Royse AG. Total arterial coronary revascularization: techniques and results in 3,220 patients. Ann Thorac Surg. 1999;68(6):2093-9. [MedLine]
3. Cameron A, Davis KB, Green G, Schaff HV. Coronary bypass surgery with internal-thoracic-artery grafts: effects on survival over a 15-year period. N Engl J Med. 1996;334(4):216-9. [MedLine]
4. Loop FD, Lytle BW, Cosgrove DM, Stewart RW, Goormastic M, Williams GW, et al. Influence of the internal-mammary-artery graft on 10-year survival and other cardiac events. N Engl J Med. 1986;314(1):1-6. [MedLine]
5. He GW. Arterial grafts for coronary artery bypass grafting: biological characteristics, functional classification, and clinical choice. Ann Thorac Surg. 1999;67(1):277-84. [MedLine]
6. Kurlansky PA, Traad EA, Dorman MJ, Galbut DL, Zucker M, Ebra G. Thirty-year follow-up defines survival benefit for second internal mammary artery in propensity-matched groups. Ann Thorac Surg. 2010;90(1):101-8. [MedLine]
7. Glineur D, D'Hoore W, Price J, Dorméus S, de Kerchove L, Dion R, et al. Survival benefit of multiple arterial grafting in a 25-year single-institutional experience: the importance of the third arterial graft. Eur J Cardiothorac Surg. 2012;42(2):284-90.
8. Martins SK, Santos MA, Tirado FHP, Martins Jr FCE, Malat HF, Jatene AD, et al. Revascularização do miocárdio com emprego de ambas artérias mamárias internas em pacientes com diabetes mellitus. Rev Bras Cir Cardiovasc. 2007;22(3):291-6. [MedLine] View article
9. Acar C, Ramsheyi A, Pagny JY, Jebara V, Barrier P, Fabiani JN, et al. The radial artery for coronary artery bypass grafting: clinical and angiographic results at five years. J Thorac Cardiovasc Surg. 1998;116(6):981-9. [MedLine]
10. Calafiore AM, Di Giammarco G, Teodori G, D'Annunzio E, Vitolla G, Fino C, et al. Radial artery and inferior epigastric artery in composite grafts: improved midterm angiographic results. Ann Thorac Surg. 1995;60(3):517-23.
11. Wei FC, Jain V, Celik N, Chen HC, Chuang DC, Lin CH. Have we found an ideal soft-tissue flap? An experience with 672 anterolateral thigh flaps. Plast Reconstr Surg. 2002;109(7):2219-26.
12. Koshima I, Yamamoto H, Hosoda M, Moriguchi T, Orita Y, Nagayama H. Free combined composite flaps using the lateral circumflex femoral system for repair of massive defects of the head and neck regions: an introduction to the chimeric flap principle. Plast Reconstr Surg. 1993;92(3):411-20. [MedLine]
13. Tatsumi TO, Tanaka Y, Kondoh K, Minohara S, Sawada Y, Tsuchida T, et al. Descending branch of lateral femoral circumflex artery as a free graft for myocardial revascularization: a case report. J Thorac Cardiovasc Surg. 1996;112(2):546-7. [MedLine]
14. Schamún CM, Durán JC, Rodríguez JM, Rodríguez M, Sanguinetti CM, Braccini G, et al. Coronary revascularization with the descending branch of the lateral femoral circumflex artery as a composite arterial graft. J Thorac Cardiovasc Surg. 1998;116(5):870-1. [MedLine]
15. Fabbrocini M, Fattouch K, Camporini G, DeMicheli G, Bertucci C, Cioffi P. The descending branch of lateral femoral circumflex artery in arterial CABG: early and midterm results. Ann Thorac Surg. 2003;75(6):1836-41. [MedLine]
16. Miller JM, Rochitte CE, Dewey M, Arbab-Zadeh A, Niinuma H, Gottlieb I, et al. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med. 2008;359(22):2324-36. [MedLine]
17. Faidutti B. Coronary artery bypass grafting with the descending branch of the lateral femoral circumflex artery used as an arterial couduit: is arteriographic evaluation necessary before its use? J Thorac Cardiovasc Surg. 1999;117(6):1229-30. [MedLine]
18. Fukuda H, Ashida M, Ishii R, Abe S, Ibukuro K. Anatomical variants of the lateral femoral circumflex artery: an angiographic study. Surg Radiol Anat. 2005;27(3):260-4. [MedLine]
19. Salm LP, Bax JJ, Jukema JW, Schuijf JD, Vliegen HW, Lamb HJ, et al. Comprehensive assessment of patients after coronary artery bypass grafting by 16-detector-row computed tomography. Am Heart J. 2005;150(4):775-81. [MedLine]
20. Marano R, Liguori C, Rinaldi P, Storto ML, Politi MA, Savino G, et al. Coronary artery bypass grafts and MDCT imaging: what to know and what to look for. Eur Radiol. 2007;17(12):3166-78. [MedLine]
21. Onuma Y, Tanabe K, Chihara R, Yamamoto H, Miura Y, Kigawa I, et al. Evaluation of coronary artery bypass grafts and native coronary arteries using 64-slice multidetector computed tomography. Am Heart J. 2007;154(3):519-26. [MedLine]
22. Ropers D, Pohle FK, Kuettner A, Pflederer T, Anders K, Daniel WG, et al. Diagnostic accuracy of noninvasive coronary angiography in patients after bypass surgery using 64-slice spiral computed tomography with 330-ms gantry rotation. Circulation. 2006;114(22):2334-41.
23. Stein PD, Beemath A, Skaf E, Kayali F, Janjua M, Alesh I, et al. Usefulness of 4-, 8-, and 16-slice computed tomography for detection of graft occlusion or patency after coronary artery bypass grafting. Am J Cardiol. 2005;96(12):1669-73. [MedLine]
24. Khan R, Rawal S, Eisenberg MJ. Transitioning from 16-slice to 64-slice multidetector computed tomography for the assessment of coronary artery disease: are we really making progress? Can J Cardiol. 2009;25(9):533-42. [MedLine]
Funding: This work was funded by FAPESP, Brazil.
Article receive on Monday, March 11, 2013
 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license