Mário IssaI; Álvaro AvezumII; Daniel Chagas DantasIII; Antônio Flávio Sanches de AlmeidaIV; Luiz Carlos Bento de SouzaV; Amanda Guerra Moraes Rego SousaVI
DOI: 10.5935/1678-9741.20130004
Stroke Stroke
CPB Cardiopulmonary bypass
Cm Centimeters
SD Standard deviation
LVEF Left ventricular ejection fraction
CI Confidence interval
Min Minutes
OR Oddis ratio
INTRODUCTION
In the United States, approximately 15 000 people a year are diagnosed with thoracic aortic aneurysm. Additionally, more than 47 thousand people per year die from aortic diseases, more than breast cancer, AIDS, homicides or car crashes, making aortic disease a silent epidemic [1].
Aneurysms and dissection constitute the main aortic diseases, which can be subjected to common principles and surgical treatment techniques. Surgical management remains a challenge in elective procedures as well as in emergencies. The decision on surgery is based on the balance between the risk and the chance of surgical aortic rupture, which can be particularly difficult in elective cases. Furthermore, among patients with thoracic aortic rupture, mortality is extremely high, reaching above 94% [2].
The primary aim of this study was to identify predictors independently associated with hospital mortality in patients undergoing surgical repair of aortic diseases. The secondary aim was to identify variables independently associated to compound clinical outcome during hospitalization (death, bleeding, neurologic complications or ventricular dysfunction).
METHODS
This research involves a cross-sectional design, with data collection and retrospective and prospective longitudinal component.
Consecutive patients with a confirmed diagnosis of ascending aortic aneurysm or type A Stanford chronic dissection were included from the record review, through retrospective data collection, beginning in January 2004. From January 2009, consecutive patients were enrolled through prospective data collection, until December 2010.
The observed sample included 257 patients whose events hospital mortality and compound clinical outcome were assessed in terms of quantitative and qualitative measures.
Inclusion criteria were patients who had undergone surgery for type A Stanford chronic dissection and ascending aortic aneurysm, involvement of the ascending aorta in all patients, commitment present or not of other aortic segments, both genders and no age limit. Exclusion criteria were patients with acute aortic dissection (of any kind), patients with aneurysms involving other aortic segments that were not ascending aorta and type B Stanford chronic dissection.
The composite clinical outcome refers to the presence of hospital mortality or neurologic complications (stroke or coma > 24 hours) or ventricular dysfunction (symptomatic heart failure and/or cardiogenic shock) or clinically relevant bleeding (presence of hemostasis review and/or need for transfusion of packed red blood cells > 3U), or that is, patients who had at least one of these events was characterized as having composite clinical outcome.
For statistical analysis, the sample of patients studied was described by absolute (n) and relative (%) frequencies of qualitative measures (factors) and summary statistics of mean, median, standard deviation (SD) and 25 and 75 percentiles of quantitative measurements (covariates).
The association between the qualitative measures and groups (mortality and composite clinical outcomes) was assessed by chi-square test or Fisher exact test. To compare quantitative measures between the two groups the nonparametric Mann-Whitney test was applied. For death after surgery were observed the Kaplan-Meier survival curves or average survival and, initially, the effects of factors assessed by the Log-Rank test and the covariates by Cox regression. The effects of factors and covariates considered statistically significant (P<0.10) or clinically relevant, were also observed, all present in a logistic regression model for multiple variables on hospital mortality and compound clinical outcome. For late death, in the long-term after surgery, multivariate were assessed by Cox regression model
The level of significance was set at 5%, or that is, differences were considered significant when P value of the tests was less than 5%. The cases in which the P value remained between 5% and < 10% suggested trends of significant effect.
This study was approved by the Research Ethics Committee of the Dante Pazzanese Institute of Cardiology in October 2010, under protocol number 3994.
RESULTS
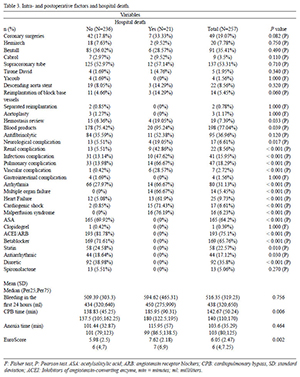
We included 257 patients, from January 2004 to December 2010, being 33.9% female, 7% of black ethnicity, with a mean age of 57.7 years. Of this population, 75% had a diagnosis of ascending aortic aneurysm and 25% of type A Stanford chronic dissection, with smoking present in 40.5%, diabetes mellitus in 13.2% and hypertension in 78, 6%. Chest pain was present in 41.6% of patients, the diagnosis of Marfan syndrome in 2.7%, and mean left ventricular ejection fraction (LVEF) assessed in 57.8%, mean EuroScore of 6.12. In this sample, 55 (21.4%) patients underwent reoperation, and in 29 (50.9%) of them only aortic valve replacement was performed in the first procedure, in nine (16.4%), surgery for myocardial revascularization, in five (9.1%), correction of acute aortic dissection, in four (7.3%), repair of aortic coarctation, in three (5.4%), aortic aneurysm, and only one case in each of the following situations: mitral valve replacement, tricuspid valvuloplasty, correction of patent ductus arteriosus, a ventricular septal defect, tetralogy of Fallot and interrupted aortic arch. The simultaneous surgery for myocardial revascularization was required in 49 (19.1%) patients.
Demographic data for all patients enrolled in this study are described in Table 1. Quantitative measures are summarized in amplitude, mean and standard deviation; qualitative measures in absolute frequency and percentage.
The Tables 2 and 7 summarize the assessments performed in this study.




DISCUSSION
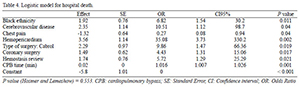
In this analysis including 257 consecutive patients who had undergone surgery during the period from January 2004 to December 2010, the overall hospital mortality rate was 8.17%. By multivariate analysis, the logistic model identified the following factors independently associated with death during hospital stay: patients of black ethnicity, associated CABG, Cabrol operation, presence of cerebrovascular disease, need for revision of hemostasis, prolonged cardiopulmonary bypass (CPB) and the presence of hemopericardium. The presence of chest pain was identified as a protective factor. Factors such as axillary cannulation site, use of antifibrinolytic, neurological complications, presence of severe arrhythmia, EuroSCORE, anoxia (min) and bleeding in the first 24 hours, by multivariate analysis, were independently associated with the occurrence of the composite clinical outcome.
Contemporary surgical series involving patients with ascending aortic disease with the use of modern techniques of grafting and methods of cerebral and myocardial protection present hospital mortality rates ranging from 1.7% to 17.1%, probably due to the heterogeneity of patient population assessed. The causes of mortality include haemorrhage, stroke and respiratory failure, and heart failure is considered the most common cause of premature death in this population [3-5].
Gazoni et al. [5] compared immediate results of elective thoracic aortic aneurysm surgery, among low-volume centers (< 40 surgeries/year) and high volume (> 80 surgeries/year). Operative mortality was 8.3% and 3.7%, respectively (P=0.02). In logistic regression analysis, low-volume centers were significantly associated with increased rates of complications and mortality (P<0.05).
In this study, 66.1% of patients were male and among hospital deaths, 57.1% were male. Gender and age had no statistically significant impact on hospital and late mortality rates, after adjustment among the different variables in the multivariate model. It should be mentioned that the age is presented as an independent risk factor for mortality in other areas of cardiovascular disease, such as acute coronary syndrome, atrial fibrillation or heart failure, but also female patients have higher mortality rates when subjected to myocardial revascularization surgery, when compared with male patients. The results of this research can be explained by two considerations: age and gender do not represent predictors independently associated with mortality in patients with aortic disease assessed in this study or due to lack of statistical power, we could not demonstrate the effect of these two variables on clinically relevant outcomes. The inclusion of additional number of patients could clarify the above considerations. Despite these differences, when all variables are entered into the logistic model, in the present study, these variables did not emerge as independent predictors of mortality.
In a study on the quality of life of patients undergoing aortic aneurysm, Lohse et al. [6] demonstrated a highly significant connection between cardiac surgeries and postoperative death (P=0.001). There was also a significant association between death and myocardial infarction (P<0.001), stroke (P=0.001), prolonged ventilation time (P<0.001), increased use of transfusion products (P=0.016) and prolonged surgical time ( P<0.001). The high incidence of myocardial infarction perioperatively was associated with a high percentage of patients with coronary artery disease. With regard to quality of life, functional class improved according to the classification of the Canadian Cardiovascular Society. One of the major impacts in reducing the quality of life of these patients was the presence of stroke, as well as prolonged hospitalization.
In a study published by Czerny et al. [7], age was not associated with increased risk of mortality and neurological injury in patients undergoing surgical repair of acute or chronic diseases of the thoracic aorta with hypothermic circulatory arrest, a finding consistent with the results of this study.
In our series, 55 (21.4%) patients underwent reoperation, and in 29 (51%) only aortic valve replacement was performed in the first procedure, in nine (16.4%), surgery for myocardial revascularization, in five (9.1%), correction of acute aortic dissection, in four (7.3%), repair of aortic coarctation, in three (5.5%), aortic aneurysm repair. Each of the following events: mitral valve replacement, tricuspid valvuloplasty, correction of patent ductus arteriosus, a ventricular septal defect, tetralogy of Fallot and aortic arch interruption occurred in only 1 case.
Although in univariate analysis for hospital mortality the prior heart surgery variable had demonstrated statistical significance (P=0.012) in multivariate analysis there was persistence of the level of significance of this variable, showing not being an independent predictor.
As shown by Estrera et al. [8], patients with type A acute dissection after previous cardiac surgery showed similar risks of poor perfusion, hypotension and cardiac tamponade. This finding suggests that adhesions formed after the operation does not eliminate the risk of cardiac tamponade due to aortic rupture. Although the results are plausible, hospital mortality was higher (31% vs. 13.8%, P <0.007) than among patients without a history of cardiac surgery.
It is recommended prophylactic replacement of the ascending aorta, although apparently normal, where there is any swelling in cases of bicuspid aortic valve. Even in the absence of significant aortic valve disease, there is often association with dilatation of the aortic root, annuloaortic ectasia and aortic dissection. After replacement of the bicuspid aortic valve, this finding is reported as a risk factor for late acute dissection and ascending aorta aneurysm [9-11].
For patients with bicuspid aortic valve normofunctioning and dilatation of the ascending aorta, the surgery is recommended for those with a diameter > 5 cm and differentiated approach for patients under surveillance in the absence of significant increase (> 0.5 cm/year). Overall survival is considered equivalent to that of a normal population considered the same age and gender. The surgery was required in approximately 10% of patients treated each year [12].
Regarding risk factors for hospital mortality, Ogeng'o et al. [13] studied the pattern of aneurysms among young black Kenyan. The patients were divided into two groups: < 40 years (group I) and > 40 years (group II). Located in the aorta, 49.6% were in group I and 68.6% in group II. Most in the abdominal aorta (64.9%), especially in the infrarenal segment. In the aortic ascending segment, below 5%, and equivalent for both groups. The gender ratio is 2.7: 1, with predominance in males. In aortic and intracranial segments, the highest incidence occurred between 31 and 40 years and between 21 and 30 at the periphery. The comorbidities in both groups were: hypertension, smoking, AIDS, alcoholism, obesity and Marfan syndrome. However, only in patients aged over 40 years, atherosclerosis and dyslipidemia were present, and diabetes mellitus was associated with peripheral and intracranial aneurysms, but not in the aorta.
The prevalence of atherothrombotic traditional risk factors varied significantly among ethnic groups. The use of medical therapy to reduce the risk was comparable among all groups. In less than 2 years of follow-up, the rate of cardiovascular mortality was significantly higher in blacks (6.1%) compared to all other ethnic groups (3.9%, P=0.01), according to Meadows et al. [14].
In previous studies, we observed that the procedure for simultaneous correction of ascending aorta and coronary artery bypass grafting can be performed safely and with good results. Ueda et al. [15] demonstrated that the incomplete coronary revascularization was identified as a risk factor for cardiovascular events (P=0.016).
In an analysis for the surgical treatment of aortic root, comparing the valved tube with preservation of the aortic valve, Dias et al. [16] demonstrated, in a consistent manner with the findings of this research, that the Cabrol's operation is one of the independent predictors of hospital and late mortality.
Murphy et al. [17], in a recent publication, found that blood transfusion in patients undergoing cardiac surgery was strongly associated with infection and ischemic postoperative morbidity, prolonged hospital stay, increased early and late mortality and increased hospital costs. Costs were substantially higher in patients who required re-exploration for bleeding, as demonstrated by Alstrom et al. [18].
As reported by Svensson et al. [19], patients undergoing surgical procedures on the ascending aorta and aortic arch, and those participating in blood conservation techniques had significantly (P<0.05) lower incidence of homologous transfusions, were extubated earlier, had lower hospitalization and were discharged in improved functional class. The multivariate analysis of 60 patients showed that the predictors of in-hospital homologous transfusion, with P values <0.05 were: age, duration of CPB and increased drainage postoperatively.
Pericardial effusion is more common in acute aortic dissection or during exacerbation of a chronic dissection. Mehta et al. [20] reported that the rate of hospital mortality for type A Stanford acute aortic dissection is high and can be predicted with the use of a clinical model incorporated into a simple risk prediction. The pericardial effusion in the clinical presentation of these patients has been identified as an independent predictor (P=0.07), as well as the cardiac tamponade complication (P<0.0001). In the model proposed predictor, the composite outcome arterial hypotension, shock and cardiac tamponade was associated with higher mortality (OR = 2.97, 95% CI 1.83 to 4.81, P<0.001).
Ranucci et al. [21] suggest that a multifactorial approach seems better suited to meet the complex reactions that lead to inflammatory lesions and activation of the coagulation cascade during cardiac surgeries. Probably, the key issue is the thrombin formation which, simultaneously, is a marker of activation of the clotting cascade and a trigger for the reactions of endothelial base, including consumption of antithrombin and protein C, activation, platelet adhesion and aggregation, expression of molecules adhesion and leukocyte adhesion and activation.
The onset of chest pain with a protective effect, reducing the risk of hospital mortality by 73% (P=0.04), can be interpreted and substantiated by epidemiological and clinical plausibility, because the waiting time for the transaction be shortened regardless of the aortic diameter, thus preventing rupture, dissection or cardiac tamponade.
With regard to compound clinical outcome in the series published by Souza & Moitinho [22], measures adopted with the use of antifibrinolytic agents, hemodilution and total replacement of the perfusate allowed to reduce the need for blood transfusion in the postoperative of cardiac surgery. Patients with CPB time greater than 120 minutes tended to need blood transfusion. The combination of surgery in elderly patients and CPB time greater than 120 minutes resulted in increased use of blood and blood products in the postoperative period. Brown et al. [23] performed a meta-analysis to compare aprotinin, epsilon-aminocaproic acid and tranexamic acid. The results included a loss of whole blood, packed red blood cell transfusion, reexploration, mortality, stroke, myocardial infarction, renal dialysis and renal dysfunction. All antifibrinolytic agents were effective in reducing blood loss and transfusion. There was no significant risks or benefits for mortality, stroke, myocardial infarction or renal failure. However, the high dose of aprotinin is associated with increased risk of statistically significant renal dysfunction.
The EuroSCORE showed clear association with the occurrence of compound clinical outcomes in this study population and should be considered an important tool in determining the risk of these patients, even being prepared in other countries, with different people and medical services.
Chertow et al. [24] assessed 42,773 patients undergoing cardiac valve surgery or CABG to determine the association between acute renal failure and need for dialysis and operative mortality. Acute renal failure occurred in 1.1% of patients. The operative mortality was 63.7% in these patients, compared to 4.3% in patients without this complication. After adjusting for factors related to comorbidities, the OR was 27, 95% CI 22-34. It was concluded that acute renal failure was independently associated with early mortality after cardiac surgery and that interventions to prevent or improve the treatment of this condition are necessary and should be implemented as soon as within this clinical-surgical scenario.
Wynne & Botti [25] described the frequency of pulmonary complications after cardiac surgery: pleural effusion (25-95%), atelectasis (17-88%), phrenic nerve palsy (30-75%), prolonged mechanical ventilation (6-58%), diaphragmatic dysfunction (2-54%), pneumonia (4-20%), diaphragmatic paralysis (9%), pulmonary embolism (0.4 to 3.2%), acute respiratory distress syndrome (0.4-2%), aspiration (1.9%) and pneumothorax (1.4%).
CONCLUSION
1. Black ethnicity, cerebrovascular disease, hemopericardium, Cabrol operation, associated CABG, hemostasis review and CPB time were independently associated with increased risk of hospital death. The presence of chest pain was independently associated with reduced risk of hospital death.
2. Use of antifibrinolytic, renal complications, pulmonary complications, EuroSCORE and prolonged cardiopulmonary bypass time were independently associated with increased risk of hospital composite clinical outcome (death, bleeding, neurologic complications or ventricular dysfunction).
ACKNOWLEDGEMENTS
The authors thank Roberta de Souza, statistics, Simone Batista da Cruz, secretary of the Division of Translational Epidemiology and Wellington Cícero de Carvalho, systems analyst.
REFERENCES
1. Hiratzka LF, Bakris GL, Beckman JA, Bersin RM, Carr VF, Casey DE Jr, et al; American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines; American Association for Thoracic Surgery; American College of Radiology; American Stroke Association; Society of Cardiovascular Anesthesiologists; Society for Cardiovascular Angiography and Interventions; Society of Interventional Radiology; Society of Thoracic Surgeons; Society for Vascular Medicine. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, American Association for Thoracic Surgery, American College of Radiology, American Stroke Association, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society of Interventional Radiology, Society of Thoracic Surgeons, and Society for Vascular Medicine. Circulation. 2010;121(13):e266-369. [MedLine]
2. Johansson G, Markström U, Swedenborg J. Ruptured thoracic aortic aneurysms: a study of incidence and mortality rates. J Vasc Surg. 1995;21(6):985-8. [MedLine]
3. Kouchoukos NT, Wareing TH, Murphy SF, Perrillo JB. Sixteen-year experience with aortic root replacement. Results of 172 operations. Ann Surg. 1991;214(3):308-18.
4. Crawford ES, Svensson LG, Coselli JS, Safi HJ, Hess KR. Surgical treatment of aneurysm and/or dissection of the ascending aorta, transverse aortic arch, and ascending aorta and transverse aortic arch. Factors influencing survival in 717 patients. J Thorac Cardiovasc Surg. 1989;98(5 Pt 1):659-73.
5. Gazoni LM, Speir AM, Kron IL, Fonner E, Crosby IK. Elective thoracic aortic aneurysm surgery: better outcomes from high-volume centers. J Am Coll Surg. 2010;210(5):855-9.
6. Lohse F, Lang N, Schiller W, Roell W, Dewald O, Preusse CJ, et al. Quality of life after replacement of the ascending aorta in patients with true aneurysms. Tex Heart Inst J. 2009;36(2):104-10. [MedLine]
7. Czerny M, Krähenbühl E, Reineke D, Sodeck G, Englberger L, Weber A, et al. Mortality and neurologic injury after surgical repair with hypothermic circulatory arrest in acute and chronic proximal thoracic aortic pathology: effect of age on outcome. Circulation. 2011;124(13):1407-13. [MedLine]
8. Estrera AL, Miller CC, Kaneko T, Lee TY, Walkes JC, Kaiser LR, et al. Outcomes of acute type a aortic dissection after previous cardiac surgery. Ann Thorac Surg. 2010;89(5):1467-74. [MedLine]
9. Nistri S, Sorbo MD, Martin M, Palisi M, Scognamiglio R, Thiene G. Aortic root dilatation in young men with normally functioning bicuspid aortic valves. Heart. 1999;82(1):19-22. [MedLine]
10. Russo CF, Mazzetti S, Garatti A, Ribera E, Milazzo A, Bruschi G, et al. Aortic complications after bicuspid aortic valve replacement: long-term results . Ann Thorac Surg. 2002;74(5):S1773-6.
11. Park CB, Greason KL, Suri RM, Michelena HI, Schaff HV, Sundt TM 3rd. Should the proximal arch be routinely replaced in patients with bicuspid aortic valve disease and ascending aortic aneurysm? J Thorac Cardiovasc Surg. 2011;142(3):602-7. [MedLine]
12. Chaturvedi N. Ethnic differences in cardiovascular disease. Heart 2003;89(6):681-6. [MedLine]
13. Ogeng'o JA, Obimbo MM, Olabu BO, Sinkeet RA. Pattern of aneurysms among young black Kenyans. Indian J Thorac Cardiovasc Surg. 2011;27(2):70-5.
14. Meadows TA, Bhatt DL, Cannon CP, Gersh BJ, Röther J, Goto S, et al; the REACH Registry Investigators. Ethnic differences in cardiovascular risks and mortality in atherothrombotic disease: insights from the Reduction of Atherothrombosis for Continued Health (REACH) registry. Mayo Clin Proc. 2011;86(10):960-7. [MedLine]
15. Ueda T, Shimizu H, Shin H, Kashima I, Tsutsumi K, Iino Y, et al. Detection and management of concomitant coronary artery disease in patients undergoing thoracic aortic surgery. Japan J Thorac Cardiovasc Surg. 2001;49(7):424-30.
16. Dias RR, Mejia OAV, Fiorelli AI, Pomerantzeff PMA, Dias AR, Mady C, et al. Análise do tratamento cirúrgico da raiz da aorta com tubo valvulado e com a preservação da valva aórtica. Rev Bras Cir Cardiovasc. 2010;25(4):491-9. [MedLine]
17. Murphy GJ, Reeves BC, Rogers CA, Rizvi SI, Culliford L, Angelini GD. Increased mortality, postoperative morbidity, and cost after red blood cell transfusion in patients having cardiac surgery. Circulation. 2007;116(22):2544-52. [MedLine]
18. Alström U, Levin LA, Stahle E, Svedjeholm R, Friberg Ö. Cost analysis of re-exploration for bleeding after coronary artery bypass graft surgery. Br J Anaesth. 2011;108(2):216-22. [MedLine]
19. Svensson LG, Sun J, Nadolny E, Kimmel WA. Prospective evaluation of minimal blood use for ascending aorta and aortic arch operations Ann Thorac Surg. 1995;59(6):1501-8. [MedLine]
20. Mehta RH, Suzuki T, Hagan PG, Bossone E, Gilon D, Llovet A, et al; International Registry of Acute Aortic Dissection (IRAD) Investigators. Predicting death in patients with acute type A aortic dissection. Circulation. 2002;105(2):200-6. [MedLine]
21. Ranucci M, Balduini A, Ditta A, Boncilli A, Brozzi S. A systematic review of biocompatible cardiopulmonary bypass circuits and clinical outcome. Ann Thorac Surg. 2009;87(4):1311-9. [MedLine]
22. Souza HJ, Moitinho RF. Estratégias para redução do uso de hemoderivados em cirurgia cardiovascular. Rev Bras Cir Cardiovasc. 2008;23(1):53-9. [MedLine] View article
23. Brown JR, Birkmeyer NJ, O'Connor GT. Meta-analysis comparing the effectiveness and adverse outcomes of antifibrinolytic agents in cardiac surgery. Circulation. 2007;115(22):2801-13. [MedLine]
24. Chertow GM, Levy EM, Hammermeister KE, Grover F, Daley J. Independent association between acute renal failure and mortality following cardiac surgery. Am J Med. 1998;104(4):343-8. [MedLine]
25. Wynne R, Botti M. Postoperative pulmonary dysfunction in adults after cardiac surgery with cardiopulmonary bypass: clinical significance and implications for practice. AM J Crit Care. 2004;13(5):384-93. [MedLine]
Article receive on Tuesday, November 27, 2012
 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license