Antonio Alceu dos SantosI; Alexandre Gonçalves de SousaII; Hugo Oliveira de Souza ThoméIII; Roberta Longo MachadoIV; Raquel Ferrari PiottoV
DOI: 10.5935/1678-9741.20130003
AF: Atrial fibrillation
CABG: Coronary artery bypass grafting
CPB: Cardiopulmonary bypass
EuroSCORE: European System for Cardiac Operative Risk Evaluation
HF: Heart failure
ICU: Intensive care unit
KF: Kidney failure
RBCT: Red blood cells transfusion
INTRODUCTION
Blood transfusions are among the greatest scientific discoveries of medical history. Since the nineteenth century they are used on a large scale worldwide. In Brazil, for example, about four million units of blood and derivatives are transfused annually [1]. The reason for this clinical practice is that the anemic patients have adverse outcomes due to reduced capacity to transport oxygen to the tissues, and hence the replacement of red cells through transfusion could prevent these deleterious outcomes [2].
Other indications, such as getting volemic expansion and blood pressure restoration, have no scientific support [3]. However, as evidenced in the worldwide medical literature, the blood transfusions have been questioned for several reasons. Firstly, we still do not have a specific, safely and effectively hemoglobin level able to set the trigger to indicate a blood transfusion [4], moreover there is a huge discrepancy of its use among physicians and hospitals [5]. Another worrying factor is the increased risk of heart failure (HF) [6], atrial fibrillation (AF), kidney failure (KF), stroke, respiratory infection, severe sepsis, and longer hospital stays [7], in the postoperative cardiac surgery, besides the malignant diseases [8], potential transmission of 68 infectious agents [9], multiple organ failure [10].
But what really has concerned the medical community according to recent studies is the mortality increase in general [11] and cardiac surgery [6,12,13]. Another important factor is the reduction of blood donations, leading to blood bank stocks below than what would be the ideal in Brazil [14] and abroad [15].
Based on these findings the use of homologous blood has been proposed more narrowly in cardiovascular surgery [16]. Therefore, it is necessary to have more evidence of this medical practice in our country. Thus, the aim of this study is to assess the 30-day and 1-year mortality associated with the red blood cells transfusion (RBCT) after coronary artery bypass grafting (CABG).
METHODS
We performed a retrospective cohort study using data from the cardiac surgery service of the Beneficência Portuguesa Hospital of São Paulo. The CABG surgeries database is powered by 14 teams, composed of 3,004 patients who were followed-up during the period of June 8, 2009 to July 26, 2010.
For the study, were considered patients undergoing CABG with or without associated procedures, age > 18 years; with no restriction on sex or race. We excluded patients underwent any other surgery, including cardiac, without CABG.
The data collection form presented a total of 243 variables. For this study were assessed nine: age, gender, comorbidities, European System for Cardiac Operative Risk Evaluation (EuroSCORE), type of surgery, packed red blood cells transfusion, length of stay (in hospital and Intensive Care Unit - ICU), acquired comorbidities after surgery, and intraoperative and postoperative complications.
We present in Tables 1 and 2 the variables assessed in this study in each group.
The patients were divided among two groups: 1) transfused, patients who received one or more units of packed red blood cells during and / or after surgery, and 2) non-transfused.
We adjusted two models for assessing the mortality in these two groups: a model for 30-day mortality and another for the 1-year mortality.
Quantitative variables were described as means and standard deviations or medians, and qualitative variables by absolute and relative frequencies. For the data analysis in both groups we used the t-Student test and Mann-Whitney nonparametric test. For checking homogeneity between ratios we used the chi-square or Fisher's exact test.
To obtain mortality prognostic factors the multivariate logistic regression model [17] was used with "stepwise" variable selection process, based on variables that presented P<0.25 in the univariate analysis. The significance level used for the tests was 5%.
This work was assessed and approved by the Ethics Committee in Research of the Hospital Beneficência Portuguesa of São Paulo, under Nº 700-11, according to the Helsinki Declaration.
RESULTS
The group of transfused patients was 1,888 (63%) and non-transfused 1,116 (37%). There was a predominance of male patients (69.9%) and whites (84.6%) with mean age of 62.2 ± 9.5 years. The survival rate was 94.4% at hospital discharge (in four cases to another hospital) and only 0.2% (seven cases) of intraoperative death.
Firstly, we found that the RBCT proved to be a predictor of 30-day mortality. The adjusted odds ratio for 30-day mortality in transfused was 2 (95% CI 1.21 to 3.31, P=0.007). The study showed a total of 129 deaths in 30 days, with 108 (84%) in transfused group and 21 (16%) in non-transfused (P<0.001). Thus, the transfusion group showed a significantly greater percentage of deaths in 30 days when compared to non-transfused (Table 3). On the other hand, we observed a mortality rate of 5.7% in the group receiving RBCT, against only 1.9% deaths among non-transfused group (P<0.001). So we have a risk reduction of 3.8%, which is equivalent to state that every 26 RBCT restrictions we avoid one death in 30 days. As shown in the Figure 1, the difference between the two groups became well evident when assessed the adjusted Kaplan-Meier survival curves of 30-day mortality (P <0.001).
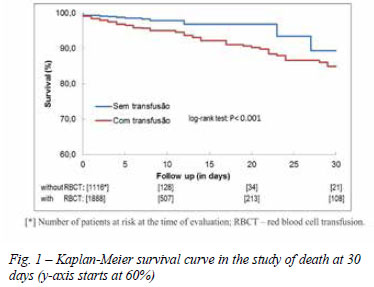
According to this study, the RBCT also showed to be a predictor of 1-year mortality. After a follow up period of 1 year deaths totaled 249 distributed in 212 (85%) in transfused group against 37 (15%) in non-transfused group (P <0.001) (Table 3). The adjusted odds ratio for 1-year mortality after red blood cell transfusion was 2.31 (95% CI 1.31 to 4.04, P=0.003). So, we also observed differences between groups for RBC transfusion in relation to the presence of death at 1 year, being significantly higher in the transfused group. In terms of percentage, we observed a 1-year mortality rate of 11.2% in the group receiving RBCT against only 3.3% in non-transfused group (P<0.001). Thus, we have a reduced risk of 7.9%, which is equivalent to say that, every 13 RBCT restrictions we avoid a death in one year.
Another data analysis in our study allows us to conclude that in both outcomes 30-day and 1-year, the non-transfused group presents significantly greater percentage of alive patients when compared to the transfused group (log-rank test P<0.001), as shown in Figure 2.
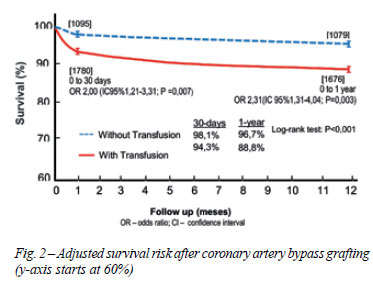
Even in low risk patients (expected mortality by EuroSCORE < 2%) we have significantly more deaths within 30 days in the transfused group (5% vs. 1%, P=0.007). In one year we also observed survival reduction in patients with low euroSCORE in transfused group (7% vs. 1%, P=0.001) (Table 4). When we separate a specific group of younger patients (<60 years) and EuroSCORE < 2%, and hence less comorbidity, we also have significantly more deaths among transfused, as shown in Table 5.
Based on these results, our study objectively shows that the RBCTs are associated with a reduction, adjusted risk, on survival in patients undergoing CABG in both 30-day and 1-year outcomes.
DISCUSSION
The real purpose of the red blood cell transfusion is to maintain adequate tissue oxygenation. Blood is a living tissue that circulates through the body delivering oxygen and nutrients to all organs. The blood has its classification into groups with the presence or absence of an antigen on the red blood cells surface. Although the ABO and Rh groups (positive and negative) are the most important, there are countless others. A transplant is the transfer of cells, tissues or organs from a living individual (donor) to another (recipient) with the aim of restoring a lost function. Thus, we can say that blood transfusions are the most common type of transplantation [18]. As it occurs with any transplant, there is an acute immune reaction and it also occurs in long term.
We have evidence to support a role of immunomodulatory effect on the transfusion with changes in the blood cells (such as a reduction in the number of circulating lymphocytes, modification in T-cells and activation of immune cells) [19]. The cardiac surgery related traumas together with RBCTs promote a noninfectious inflammatory response reflected by an increase in the inflammatory mediator concentrations. In these circumstances cytokine levels are very high. Other limitations in respect to the blood transfusions is related to the stored blood that have a reduced tissue oxygen delivery and as the storage time increases, the red cells undergo to cellular, biochemical, hormonal and inflammatory structural changes, resulting in the inability of the stored blood to perfuse the microcirculation [20]. Recently, a study reported an association between blood transfusions and the pulmonary coagulation activation, as well as intraoperative thromboembolic complications [11]. These and other still unknown factors, respond largely the negative outcomes associated with blood transfusions, specifically the early mortality of patients undergoing CABG.
This study showed that the death risk presented a most influential early impact of the blood transfusion for the first 30 days after surgery, as we can see by comparing the survival curves (Figures 1 and 2). When assessing the graphs it is possible to observe an association between the RBCT and reduced survival, statistically and significantly different, from those who did not receive a RBCT at 30 days (P<0.001). The RBCT effect on the risk of early mortality (first 6 months after surgery) was also found by Koch et al. [13]. Thus, though, an analysis of survival curve in one year is also possible to observe higher mortality among the transfused group (Figure 2).
The long-term mortality related to red blood cell transfusions is still speculative. Some of the mechanisms responsible for this effect would be the systemic inflammatory response and the immunomodulation [19]. But, we also can say that comorbidities (such as: HF, AF, stroke, KF, infectious agents transmission, pulmonary thromboembolism, and other malignancies) [6-11], caused by the transfusions also influence the late mortality.
The higher early mortality increase is believed to be related with cardiopulmonary dysfunction, and as well as the infections increase, higher inflammatory response, and the transfusion immunomodulatory effect.
With respect to different response we found with a large increase in short term mortality, and then, a sustained increase in long-term mortality in transfused patients, suggesting two distinct processes. One of these processes is the hypothesis that a transfused patient is sicker. However, the increased mortality, also in long term, discards the argument that transfusion is an indicator of hospitalized patients (Figure 2). Other information from our study, contrary to the idea that the higher mortality after a RBCT is due to the patient's more severe condition, was provided by the EuroSCORE calculation, a simple and objective index, but it has been shown as a satisfactory predictor of mortality in patients undergoing CABG [21]. When we have an EuroSCORE < 2%, the mortality is low, but in our study when we provide RBCT the death risk increased significantly in both at 30 day and 1-year mortality (Table 4). Interestingly, when we have patients younger than 60 years and an EuroSCORE < 2%, and so with fewer comorbidities and/or aggravating risk factors for heart surgery, we had no deaths in the non-transfused group (Table 5), unlike the group that received RBCT, where we had a significant mortality, in both early and late period. It is known that intraoperative allogeneic transfusion, which is usually based on hemoglobin levels and not on the patient's clinical status (symptoms and signs), is also a risk factor for the increased mortality [12]. Thus, we can state that the blood product transfusion is not necessarily an indicator factor of sicker patient, but it is an independent factor of early and late mortality.
The potential adverse influence of blood transfusions on early and long term survival is indeed alarming, as was also demonstrated by Kuduvalli et al. [22]. Our findings were also evidenced by Michalopoulos et al. [23], which reported an independent association of blood transfusions with early and late mortality after CABG.
Engoren et al. [12] also studying the effects of blood transfusions in patients undergoing CABG alone, also reported that transfused patients had twice the mortality of non-transfused. Even after the adjustment for comorbidities and other factors, the blood transfusion was still independently associated with an increase of 70% in mortality.
Other authors also reported an association between RBCT and increased mortality in a series of clinical scenarios, both in cardiac and general surgery [6,11,12].
There is a global real need in making more decisions based on evidence, in relation to the RBCTs. The proposal to maintain certain hemoglobin level via blood transfusions has no strong support in the medical literature. Several randomized and controlled studies have shown that lowering the hemoglobin threshold for recommending a blood transfusion in cardiac surgery [16] and in critically ill patients [4] do not adversely affect patient outcome.
In 2010 was published a large study that identified a dramatic variability on RBCT rates (7.8% to 92.8%) in patients undergoing CABG in several hospitals [5], demonstrating thereby that is often the physician and not the patient who does not tolerate low hemoglobin and/or hematocrit levels during or after a surgery [24]. Researchers have found that a hematocrit level between 17% and 21% during CABG together with moderate hypothermia, are well tolerated and have no adverse impact on the outcomes. In our study, even within a single center (hospital) we also recorded a wide variation in blood transfusion rates. Among the fourteen teams that perform CABG, the team who most indicated transfusion had 85.7% of their patients receiving RBCT and that who less indicated transfusion had 54.1%. This demonstrates that blood products are still used on a large scale in our country, hence the relevance of this study that adds itself to several others in highlighting the adverse effects of this medical practice.
Various techniques and strategies to reduce the blood use have been studied; one of these is the blood products during cardiopulmonary bypass (CPB) associated with a reduced hydric balance, proposed by Souza & Braile [25]. Other proposed measures are the use of antifibrinolytic agent, aminocaproic acid [26], minimum use of CPB, especially in relation to its duration [27]. Others relevant care include preoperative patient meticulous assessment, implementation of intraoperative and postoperative measures for reducing blood loss and transfusion, active involvement of the surgeon, physician, anesthesiologist and intensivist, and as well as the use of algorithms for patients assessment and treatment, can significantly reduce transfusion need and prove to be lifesaving in cases of serious bleeding. This was evidenced by previous studies that reported a reduction of up to 50% in transfusion rates with the implementation of the cardiac surgery multidisciplinary protocols [28].
The tolerance of anemia in the perioperative period is one of the three main pillars of the blood conservation, and it does not increase the risk of complications and death in cardiac surgery [29]. Another important pillar is to optimize the erythrocyte mass by stimulating the red blood cell production through specific medication (ferrous sulfate, folic acid, vitamin B12, and erythropoietin). The third pillar, also equally important, involves the blood loss reduction by using all available resources such as electrocautery, argon beam coagulation, heparin-coated circuits, leukocyte filter, anti-fibrinolytic drugs, buffer fibrin glue, hemostatic agents, normothermia, induced hypotension, meticulous hemostasis, phlebotomy, acute normovolemic hemodilution [27], and especially the intraoperative cell salvage (cell saver).
The allogeneic blood is expensive, difficult to obtain, transport and store. The local and global shortages are imminence [14,15]. Currently, there are no demonstrated real benefits of blood transfusions, and adverse effects are increasingly described. Thus, alternatives to this procedure provide many advantages, and its use is likely to improve the outcomes, as the safer and more effective agents are developed. Therefore, they should be encouraged worldwide.
There are some limitations in our study. The first is that by being a retrospective database study, by its nature, we can only find associations between variables and outcomes, without however showing the causality. A second limitation is the fact that there was not a well-established rule for transfusion in both intra and postoperative period, and the transfusion triggers were dependent on the physician responsible for the patient. Another limitation is that we use all-cause of mortality as a final outcome, not differentiating causes of cardiac and non-cardiac death. A final limitation is that we have no data to differentiate transfusions, both from intraoperative data to the postoperative period, since intraoperative transfusions, especially in surgery with CPB, are more triggered by levels of hemoglobin or hematocrit rather than patient's clinical status, and having added this parameter in our study probably would reinforce our conclusion.
CONCLUSION
The red blood cell transfusion after coronary artery bypass grafting surgery increases significantly the 30 day and 1-year mortality, even after the adjustment for comorbidities and other factors. Other therapeutic options and strategies for autologous blood management and conservation should be encouraged to reduce the transfusion need of blood products.
REFERÊNCIAS
1. Boletin de Hemovigilância Nº 2-ANVISA-2009;P:7. Accessed in 01/July/2012: http://portal.anvisa.gov.br/wps/wcm/connect/3a17a980474585998f47df3fbc4c6735/boletim_hemo_2009.pdf?MOD=AJPERES.
2. Practice Guidelines for blood component therapy: a report by the American Society of Anesthesiologists Task Force on Blood Component Therapy. Anesthesiology. 1996;84(3):732-47. [MedLine]
3. American Society of Anesthesiologists Task Force on Perioperative Blood Transfusion and Adjuvant Therapies. Practice guidelines for perioperative blood transfusion and adjuvant therapies: an updated report by the American Society of Anesthesiologists Task Force on Perioperative Blood Transfusion and Adjuvant Therapies. Anesthesiology. 2006;105(1):198-208. [MedLine]
4. Napolitano LM, Kurek S, Luchette FA, Corwin HL, Barie PS, Tisherman SA, et al; American College of Critical Care Medicine of the Society of Critical Care Medicine; Eastern Association for the Surgery of Trauma Practice Management Workgroup. Crit Care Med. 2009;37(12):3124-57. [MedLine]
5. Bennett-Guerrero E, Zhao Y, O'Brien SM, Ferguson TB Jr, Peterson ED, Gammie JS, et al. Variation in use of blood transfusion in coronary artery bypass graft surgery. JAMA. 2010;304(14):1568-75. [MedLine]
6. Murphy GJ, Reeves BC, Rogers CA, Rizvi SI, Culliford L, Angelini GD. Increased mortality, postoperative morbidity, and cost after red blood cell transfusion in patients having cardiac surgery. Circulation. 2007;116(22):2544-52. [MedLine]
7. Dorneles CC, Bodanese LC, Guaragna JCVC, Macagnan FE, Coelho JC, Borges AP, et al. O impacto da hemotransfusão na morbimortalidade pós-operatória de cirurgias cardíacas. Rev Bras Cir Cardiovasc. 2011;26(2):222-9. [MedLine] Visualizar artigo
8. Silverman RH, Nguyen C, Weight CJ, Klein EA. The human retrovirus XMRV in prostate cancer and chronic fatigue syndrome. Nat Rev Urol. 2010;7(7):392-402. [MedLine]
9. Stramer SL, Hollinger FB, Katz LM, Kleinman S, Metzel PS, Gregory KR, et al. Emerging infectious disease agents and their potential threat to transfusion safety. Transfusion. 2009;49(Suppl 2):1S-29S. [MedLine]
10. Moore FA, Moore EE, Sauaia A. Blood transfusion. An independent risk factor for postinjury multiple organ failure. Arch Surg. 1997;132(6):620-4.
11. Glance LG, Dick AW, Mukamel DB, Fleming FJ, Zollo RA, Wissler R, et al. Association between intraoperative blood transfusion and mortality and morbidity in patients undergoing noncardiac surgery. Anesthesiology. 2011;114(2):283-92. [MedLine]
12. Engoren MC, Habib RH, Zacharias A, Schwann TA, Riordan CJ, Durham SJ. Effect of blood transfusion on long-term survival after cardiac operation. Ann Thorac Surg. 2002;74(4):1180-6. [MedLine]
13. Koch CG, Li L, Duncan AI, Mihaljevic T, Loop FD, Starr NJ, et al. Transfusion in coronary artery bypass grafting is associated with reduced long-term survival. Ann Thorac Surg. 2006;81(5):1650-7. [MedLine]
14. Novaretti MCZ. Importância dos carreadores de oxigênio livre de células. Rev Bras Hematol Hemoter. 2007;29(4):394-405.
15. Sojka BN, Sojka P. The blood donation experience: self-reported motives and obstacles for donating blood. Vox Sang. 2008;94(1):56-63. [MedLine]
16. Hajjar LA, Vincent JL, Galas FR, Nakamura RE, Silva CM, Santos MH, et al. Transfusion requirements after cardiac surgery: the TRACS randomized controlled trial. JAMA. 2010;304(14):1559-67. [MedLine]
17. Hosmer Jr. DW, Lemeshow S. Applied logistic regression. New York: John Wiley & Sons;1989. p.307.
18. Berkow R, Fletcher AJ, Bondy PK, Faling LJ, Feinstein AR, Frenkel EP, et al. Transfusão de sangue. Manual Merck. 15ª ed. 1987:p.1246.
19. Blajchman MA. Immunomodulation and blood transfusion. Am J Ther. 2002;9(5):389-95. [MedLine]
20. Spiess BD. Red cell transfusions and guidelines: work in progress. Hematol Oncol Clin North Am. 2007;21(1):185-200. [MedLine]
21. Moraes Neto F, Duarte C, Cardoso E, Tenório E, Pereira V, Lampreia D, et al. Avaliação do EuroSCORE como preditor de mortalidade em cirurgia de revascularização miocárdica no Instituto do Coração de Pernambuco. Rev Bras Cir Cardiovasc. 2006;21(1):29-34. Visualizar artigo
22. Kuduvalli M, Oo AY, Newall N, Grayson AD, Jackson M, Desmond MJ, et al. Effect of peri-operative red blood cell transfusion on 30-day and 1-year mortality following coronary artery bypass surgery. Eur J Cardiothorac Surg. 2005;27(4):592-8. [MedLine]
23. Michalopoulos A, Tzelepis G, Dafni U, Geroulanos S. Determinants of hospital mortality after coronary artery bypass grafting. Chest. 1999;115(6):1598-603. [MedLine]
24. Senay S, Toraman F, Karabulut H, Alhan C. Is it the patient or the physician who cannot tolerate anemia? A prospective analysis in 1854 non-transfused coronary artery surgery patients. Perfusion. 2009;24(6):373-80. [MedLine]
25. Souza DD, Braile DM. Avaliação de nova técnica de hemoconcentração e da necessidade de transfusão de hemoderivados em pacientes submetidos à cirurgia cardíaca com circulação extracorpórea. Rev Bras Cir Cardiovasc. 2004;19(3):287-94. Visualizar artigo
26. Benfatti RA, CarliAF, Silva GVR, Dias AEMAS, Goldiano JA, Pontes JCDV. Influência do ácido épsilon aminocapróico no sangramento e na hemotransfusão pós-operatória em cirurgia valvar mitral. Rev Bras Cir Cardiovasc. 2010;25(4):510-5. [MedLine] Visualizar artigo
27. Souza HJB, Moitinho RF. Estratégias para redução do uso de hemoderivados em cirurgia cardiovascular. Rev Bras Cir Cardiovasc. 2008;23(1):53-9. [MedLine] Visualizar artigo
28. Van der Linden P, Dierick A. Blood conservation strategies in cardiac surgery. Vox Sang. 2007;92(2):103-12. [MedLine]
29. Moskowitz DM, McCullough JN, Shander A, Klein JJ, Bodian CA, Goldweit RS, et al. The impact of blood conservation on outcomes in cardiac surgery: is it safe and effective? Ann Thorac Surg. 2010;90(2):451-8. [MedLine]
Article receive on sexta-feira, 29 de junho de 2012
 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license