Objective: To study morphofunctional alterations induced by brief pressure increases in human saphenous veins utilized in coronary artery bypass grafting.
Method: Saphenous veins of 20 patients undergoing coronary artery bypass grafting, were distributed into four experimental groups, control, 100 mmHg, 200 mmHg and 300 mmHg, and submitted to pressure distention over 15 seconds using Krebs solution. The evaluation included CD34 immunohistochemistry and an In vitro vascular reactivity study in organ chambers.
Results: The main experimental findings were 1) From pressures of 200 mmHg there was a tendency to reduce the CD34 expression which became statistically significant at 300 mmHg; 2) There was no impairment of the contraction and relaxation as evidenced by in vitro vascular reactivity tests.
Conclusion: Although vascular reactivity impairment was not demonstrated in vitro, the CD34 expression, measured by imunohistochemistry, shows there is endothelium dysfunction at pressures of 300 mmHg.
Objetivo: Estudar as alterações morfofuncionais induzidas por pressão de distensão, em veias safenas humanas utilizadas para revascularização do miocárdio.
Método: Foram estudadas veias safenas de 20 pacientes, distribuídas em quatro grupos experimentais: controle, 100, 200 e 300 mmHg, submetidos a distensões pressóricas com solução de Krebs por 15 segundos. A metodologia utilizada incluiu: 1) Imunohistoquímica do CD34; 2) Estudo in vitro da reatividade vascular em câmaras de órgãos.
Resultados: Os principais achados experimentais foram: 1) A partir da pressurização com 200 mmHg, observou-se uma tendência à diminuição da expressão do CD34, tornando-se estatisticamente significante com 300 mmHg; 2) Não houve comprometimento da contratilidade e dos relaxamentos estudados in vitro.
Conclusões: Embora o estudo in vitro não tenha demonstrado comprometimento da reatividade vascular das veias estudadas, o estudo imunohistoquímico do CD34 mostrou que existe disfunção endotelial com pressurizações de 300 mmHg.
INTRODUCTION
It is well known that the patency of saphenous vein grafts is much worse than arterial grafts. This is partially due to the fact that the wall of the normal saphenous vein has different structural and functional characteristics, which may be affected by high pressure distension in the preparation phase of the vein and after its implantation in the arterial system. The saphenous vein is extensively used as a graft in the arterial system. This, consequently, exposes the venous wall to high pressure distension. Moreover, during preparation, varying degrees of distension may be caused, which may even be extremely high. These abnormal pressures might, at least in part, be responsible for pathological alterations of the wall of the saphenous vein graft both in the immediate and late postoperative periods.
The utilization of arterial pressures to expand the vein has been proposed to assess the capacity of saphenous veins as coronary grafts. This pressure would not negatively affect the vasodilator component of endothelium function, but may cause morphological changes in the vascular wall. Excessive distension produces deep structural and biochemical alterations of the endothelium that are not correlated with short-term functional changes of these cells, but can predispose the vein to long-term functional alterations by initiating proliferation and atherosclerosis of the tunica intima. These data may be clinically and experimentally important in the biological and pharmacological aspects of endothelial function and for the use of the saphenous vein as an arterial graft.
It is practically impossible to predict how mechanical stimulation of nitric oxide release, as in shear stress, might be affected by distension of the saphenous vein due to pressure. Thus, it is hard to understand why, when using saphenous veins of similar qualities, some function for more than 15 years while others have impaired flows in a much shorter time span.
The above data inspired the current study, which aims at studying possible endothelial dysfunction caused by pressure distension using: 1) Immunohistochemistry with CD34 as a marker of endothelial dysfunction and 2) an in vitro study of the stages of the nitric oxide release pathway in organ chambers, correlating the results with increasing distension pressures.
METHOD
Saphenous vein segments from the region near to the internal malleolus of 20 male and female patients, who were submitted to coronary artery bypass grafting, were obtained in the surgical room. These venous segments were gently released from surrounding tissues and their collateral branches were ligated. Following this, they were immediately immersed in normothermic oxygenated saline solution with the following millimolar composition: NaCl 118.3; KCl 4.7; MgSO4 1.2; KH2PO4 1.22; CaCl2 2.5; NaHCO3 25.0 and glucose 11.l (control solution). The veins were, at no moment, submitted to distension by the injection of saline solution during their harvesting. Veins without macroscopic evidence of varicosities were selected, but the diameter and wall thickness and the macroscopic observation of vasospasms were not considered.
All the procedures were reviewed and approved by the Research Ethics Committee of Hospital das Clinicas of the Medicine School in Ribeirão Preto, University of São Paulo, Brazil.
Standardization of pressure distension
To standardize the pressure distension, the saphenous vein segment was connected to a three-way tap, with two outlets connected to a mercury manometer and to an insufflator used for balloon angioplasty. This insufflator allows a very accurate pressure increase for a fixed period of time. As insufflation pressures of angioplasty balloons are extremely high, the manometer of the insufflator has both ATM and PSI pressure scales. Hence, one outlet of the three-way tap was connected to a mercury column manometer to measure the experimental pressures (100, 200 and 300 mmHg). To apply pressure, the free end of the saphenous vein segment was clamped (Figure 1). All preparations were performed by a single researcher. The vein segments were divided into four experiment groups: Group 1 - Control; Group II - Distension with Krebs solution to 100 mmHg for 15 seconds; Group III - Distension with Krebs solution to 200 mmHg for 15 seconds and Group IV - Distension with Krebs solution to 300mmHg for 15 seconds.

Fig. 1 - Standardization of the pressure exerted on the saphenous veins
The samples were blinded so that it was impossible for investigators to discover which group any determined sample belonged to. Identification of the groups of samples was only made after the experimental observations during quantification and analysis of possible changes induced by pressure distension.
Immunohistochemistry for CD34
For these studies, standard staining techniques were used with samples embedded in paraffin (Novostain Universal Quick Kit) after incubating saphenous vein tissues with monoclonal antibodies of constitutional and inducible NO synthesis. The antibody used was CD34 (Santa Cruz ICO115 sc-7324, dilution PBS 7.4/BSA 1/20). CD34 is a recognized histochemical marker for endothelial dysfunction.
Study of the vascular reactivity in organ chambers
Organ chambers are a double chamber made of glass where water, at a fixed temperature of 37ºC, constantly circulates in a closed circuit. Additionally, there is a reservoir containing a constant volume of 25 mL in which the vascular rings are suspended and drugs are administered. This reservoir was filled with a modified Krebs solution and connected to vacuum suction and ventilation system, allowing studies in normoxic or hypoxic conditions. The Krebs solution can be replaced whenever required, combining suction with the reintroduction of fresh solution which is stored in a glass tower, which, similarly to the organ chambers is also maintained at a temperature of 37ºC and with continuous ventilation.
Venous ring segments were suspended using stainless steel loops through their lumen. Before starting experiments using pharmacological agents, the vessels are submitted to their optimal stretch-tension point using a micrometrical system. To determine this point, the vessels were progressively stretched up to 2.5g. The isometric tension was registered using a sensitive system of strain gauge transducers (Statham UC2) individually coupled to the vascular rings and linked to an eight-channel recorder (Gould, Cleveland, Ohio) (Figure 2).
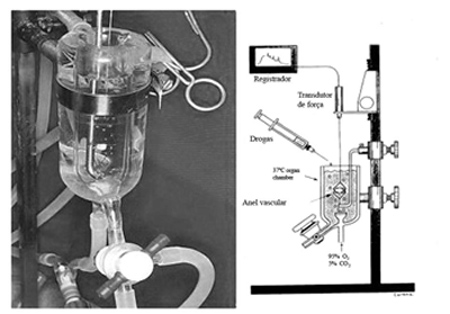
Fig. 2 - Detail of an isolated "organ chamber" and its design
As the aim of the current work was to study only the action of nitric oxide, excluding the participation of elements of the cyclooxygenase pathway, after reaching the stretch-tension point, the vessels were submitted to the action of indomethacin.
To study the contractile response of the saphenous vein segments, phenylephrine, prostaglandin F2á, noradrenalin and potassium chloride were utilized. To investigate the endothelium-dependent relaxation, three drugs were chosen: Adenosine Diphosphate (ADP), a platelet product that acts by stimulating purine receptors in the endothelial cell membrane; Calcium ionophore A23 187, which induces endothelium-dependent vasodilation independent of receptor stimulation and demonstrates that the endothelial cell maintains its capacity to produce nitric oxide and sodium nitroprussiate, which is a NO donor that acts directly on the vascular smooth muscle, with the function of testing endothelium-independent vascular relaxation.
Data analysis
Results are presented as means ± standard error as their distribution was not normal. In all experiments, n represented the number individuals from which vascular rings were attained. In phenylephrine-contracted rings, the responses are expressed as percentage of alterations compared to the maximum stable contraction. For relaxation, the negative logarithm was calculated for the effective 2-molar concentration of the agonist that caused 50% inhibition (CI 50) of the contraction obtained by prostaglandin F2á. These values were used to construct dose-response curves. Analysis of variance (ANOVA) and the Student t-test were utilized in the statistical analysis of paired and non-paired observations. The values were considered significant when the p-value was less than 0.05.
RESULTS
Immunohistochemistry tissue expression of CD34
No significant differences were observed between the control group and the group of veins pressurized to 100 mmHg. For pressures of 200 mmHg and higher, the CD34 expression tended to reduce, with statistical significance being observed at pressures of 300 mmHg (Figures 3 & 4).
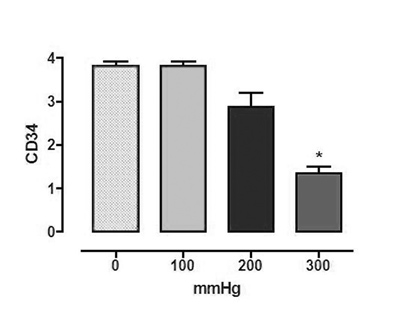
Fig. 3 - Immunohistochemistry of CD34 (n=20, ANOVA, p<0.05). The asterisk indicates statistically significant difference. Semi-quantitative analysis (the expression was graduated from + to ++++ by a single professional who was blinded to the experimental groups
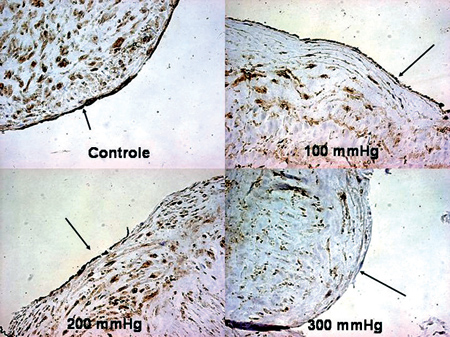
Fig. 4 - Immunohistochemistry of CD34. The arrows indicate a reduction in the expression with pressure (400x)
Contractile responses
1) Phenylephrine: As an isolated datum, a higher contractile response was observed in veins submitted to pressures of 100 mmHg which was significantly greater in respect to the control. 2) Prostaglandin F 2á: With the exception of the isolated datum of a significant reduction in contractility of veins pressurized to 300 mmHg, no other significant differences were observed among contractile responses induced by prostaglandin F 2á. 3) Noradrenalin: No significant differences were observed among the contractile responses induced by noradrenalin. 4) Potassium chloride: Although there was a clear tendency for the contractile responses induced by KCl to drop as the pressures increased, these differences were not statistically significant (Figure 5).
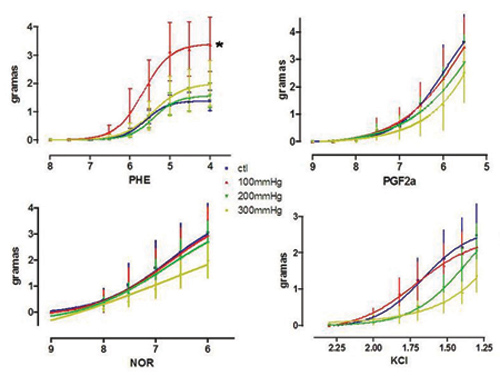
Fig. 5 - Dose-response curve to increasing concentrations of phenylephrine, prostaglandin F2á, noradrenalin and potassium chloride (n=6, ANOVA, p<0.05, only for contractions of the saphenous veins distended to 100 mmHg, for the other parameters p>0.05). The * indicates statistical difference
1) Adenosine diphosphate (ADP): No significant differences were observed for the endothelium-dependent relaxation response due to ADP (receptor-dependent). 2) Calcium ionophore: No significant differences were verified for the endothelium-dependent relaxation response caused by calcium ionophore and 3) Sodium nitroprussiate (SNP): No significant differences were demonstrated for the endothelium-independent relaxation response caused by SNP (Figure 6).
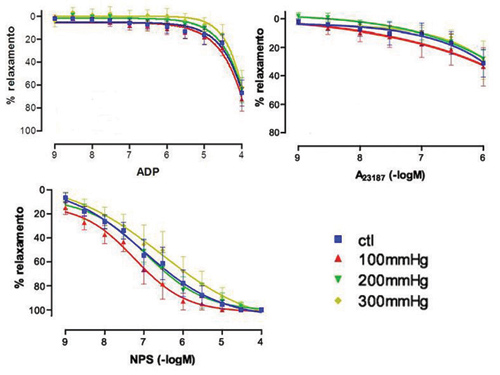
Fig. 6 - Dose-response curve to growing concentrations of adenosine diphosphate (receptor-dependent relaxation), calcium ionophore (receptor-independent relaxation) and sodium nitroprussiate (endothelium-independent relaxation) - (n=6, ANOVA, p>0.05)
The saphenous vein is still the most commonly used graft in coronary artery bypass grafting surgery together with the pedicled internal thoracic artery, even with the advantages of arterial grafts being well known. These advantages include better long-term results as arterial grafts are adjusted to the high arterial blood pressures. Adaptation depends on the paracrine function of the endothelium, which, due to the production of vasodilator and antiplatelet substances, prevents spasms, thrombosis and proliferation, and even protects against the mechanisms of atherosclerosis.
This functional inferiority of saphenous veins has been widely studied, with great importance being attributed to possible traumatic and functional lesions during harvesting and preparation for coronary artery bypass grafting. Once harvested, the vein is cannulated and a low pressure is applied to identify and ligate collateral vessels, which were not previously identified. The patency rates may be related to vein lesions during its removal and preparation [1-3].
During the 1960s and 1970s, morphological lesions were clearly established that not only involved the endothelium, but the other layers that form the venous wall. For these studies, vein pressurization was a prohibitive practice that was not observed in the evolution of coronary artery bypass grafting surgery. Thus, a great challenge emerged, which was to discover the relationship between morphological lesions caused by high pressures and functional lesions. In other words, does functional barotrauma occur in saphenous veins under pressure?
In the 1990s, working in the Mayo Clinic (Rochester, MN, USA), we saw that, in vitro studies using organ chambers, infusion of crystalloid cardioplegic solution (at pressures higher than 600 mmHg) did not induce endothelial dysfunction due to impairment of NO release in normal canine coronary arteries [4].
In perhaps the best designed study yet [5], which motivated this investigation, aiming at studying the relationship between the intensity of distension pressure and structural, biochemical and functional lesions of the saphenous vein wall, segments of this vessel were stretched using pressures of from 100 to 300 mmHg with the vascular reactivity being studied in organ chambers. In additional to functional studies, the structural lesions were evaluated using scanning microscopy and by the immunohistochemical identification of eNOS. Saphenous vein segments distended by pressures of 100 mmHg maintained their capacity to respond to potassium chloride (90 mmo1/L) and to phenylephrine (10-6 mo1/L), but those exposed to pressures of 300 mmHg lost their reactivity to these agents. Veins distended at 300 mmHg also suffered impairment of endothelium-dependent relaxation due to acetylcholine (10-10 - 10-6 mo1/L) and bradykinin (10-10 - 10-6 mo1/L). Quantitative studies of endothelial structural lesions were apparent after pressures of 300 mmHg were employed, but residual endothelial cells showed strong positive immunohistochemistry staining for eNOS. In contrast to veins submitted to 100 mmHg pressures, veins pressurized to 300 mmHg presented with large areas of denuded endothelium. This study concluded that distension of saphenous veins by pressures equivalent to high arterial pressures results in structural and biochemical changes to the endothelium "that are not accompanied by immediate functional changes". In other words, the endothelium may have great functional reserve.
During this investigation, information was being collected from publications about the effects of mechanical forces on endothelial function. The arterial bed, which consists of large-caliber muscular and elastic vessels, as well as arterioles and precapillary vessels, is constantly exposed to hemodynamic forces that vary immensely in magnitude, frequency and direction. These forces consist of pressures acting perpendicularly on the vascular wall, the cyclic effort and shear stress acting parallel to the vessel walls, forming a shear frictional force on the surface of the endothelium. When the saphenous vein is inserted in the arterial circulation, it starts to suffer the action of these physical phenomena. Added to these types of forces are the possible effects of traction on the functioning of the saphenous vein. But, the design of the current research focused on the hydrostatic pressure caused by infusion of Krebs saline solution during the harvesting and preparation of the saphenous vein. The studied segments were all from the initial portion of the saphenous vein close to the internal malleolus of the right leg. These veins were not handled much; there were no varicosities or macroscopic inflammatory reactions. They were not initially subjected to pressure nor exposed to any vasodilator agents. We believe that twenty veins from different patients are, even under the strictest criticism, a reasonable number to attenuate the extreme variability of vessels from patient to patient. There is a possible bias related to always using of the initial portion of the vein with the possibility of differences in the endothelium structure along the vein. The existence of a possible bias was abated in a recent publication from the Texas Heart Institute (Houston, TX, USA) that, using in vitro studies of saphenous veins submitted to the vasodilator action of acetylcholine (endothelium-dependent) and sodium nitroprussiate (endothelium-independent), did not identify functional differences between proximal and distal segments [6].
The main experimental finding of the current research was that, from pressures of 200 mmHg there is a tendency to reduce the CD34 expression, which becomes statistically significant at pressures of 300 mmHg but without impairment of either the contractility or relaxation as seen in "in vitro" studies using organ chambers.
Several studies have demonstrated the reactivity of CD34 antibodies in capillaries of different tissues including tumors. Practically all CD34 antibodies that we know bind to the vascular endothelium, making this an excellent marker of endothelial function. However, not all in vivo endothelial cells are positive for CD34. Moreover, even in CD34-positive tissues, staining varies immensely. Endothelial cells of large veins and arteries are negative for CD34, as is the endothelial surface of the placenta and hepatic sinusoids [7].
The CD34 antibody has proven to be an excellent marker of the human saphenous vein endothelium. A clear reduction in the CD34 antibodies occurred with pressure demonstrating endothelial dysfunction. This trend initially becomes apparent at pressures of 200 mmHg with statistical significance seen at 300 mmHg. These findings show that pressures of up to 100 mmHg do not cause endothelial dysfunction and that distensions at high pressures are directly associated to endothelial dysfunction in human saphenous veins.
It is important to stress the well-known difficulties of in vitro studies on venous vascular reactivity. This difficulty has already been reported in experimental animal studies however, it is much greater when related to adult human veins. This complexity led us to study only veins without mechanical removal of the endothelium that, in most cases, practically abolishes the contractility of the vessel. Hence, we did not use acetylcholine in the study of endothelium-dependent relaxation with stimulation from membrane receptors. Numerous studies have been performed so, eventually, we decided to adopt ADP as the pharmacological agent to induce endothelium-dependent relaxation.
The only statistically significant differences are those that correspond to an increase in the contractility due to phenylephrine in veins submitted to 100 mmHg pressures and the reduction in contractility due to the actions of prostaglandin F2á and potassium chloride. Together, in particular when taking into account that there is no impairment in the endothelium-dependent (ADP and calcium ionophore) and -independent (NPS) relaxation, we chose to consider the significant changes in contractility as being temporary. Thus, we conclude that the in vitro pharmacological studies were not sufficiently sensitive to prove that pressures of up to 300 mmHg functionally impair human saphenous veins.
CONCLUSIONS
From the data obtained from the immunohistochemistry of CD34 alone, we concluded, without any doubt, that extensive distension of the saphenous vein results in impairment of the endothelial function. It is thus possible to conclude that endothelial dysfunction occurs, without evident impairment of the vascular reactivity in "in vitro" studies and that this degree of dysfunction is less evident with pressures of up to 100 or 120 mmHg. From this we can speculate that, although the experimental period was short, there is no impairment of carefully removed and pressurized saphenous veins; surgical management, in some way, causes a kind of "molecular scaring", that may impair, over different time periods, the patency of coronary venous grafts.
It is important to stress that the current study only took into account the effects of the hydrostatic pressure, without considering the other two forces recognized as participating in vascular reactivity. As cited in a review of literature previously published in this journal [8], each type of force that is exerted on the vascular wall is associated to an expression of different genes, which makes this subject even more complex, deserving the aforementioned title of a "never-ending story".
REFERENCES
1. Soyombo AA, Angelini GD, Bryan AJ, Newby AC. Surgical preparation induces injury and promotes smooth muscle cell proliferation in a culture of human saphenous vein. Cardiovasc Res. 1993;27(11):1961-7. [
MedLine]
2. Bonchek LI. Prevention of endothelial damage during preparation of saphenous veins for bypass grafting. J Thorac Cardiovasc Surg. 1980;79(6):911-5. [
MedLine]
3. Angelini GD, Breckenridge IM, Williams HM, Newby AC. A surgical preparative technique for coronary bypass grafts of human saphenous vein which preserves medial and endothelial functional integrity. J Thorac Cardiovasc Surg. 1987;94(3):393-8. [
MedLine]
4. Évora PRB, Pearson PJ, Schaff HV. Cardioplegia cristalóide, barotrauma e função endotelial. Considerações experimentais. Rev Bras Cir Cardiovasc. 1996;11(2):115-22.
5. Chester AH, Buttery LD, Borland JA, Springall DR, Rothery S, Severs NJ, et al. Structural, biochemical and functional effects of distending pressure in the human saphenous vein: implications for bypass grafting. Coron Artery Dis. 1998;9(23):143-51.
6. Golbasi I, Tasatargil A, Aksoy NH, Sadan G, Karasu E, Turkay C, et al. A functional and histopathological comparison of proximal and distal saphenous vein contractility and morphology. Tex Heart Inst J. 2005;32(3):287-93. [
MedLine]
7. Fina L, Molgaard HV, Robertson D, Bradley NJ, Monaghan P, Delia D, et al. Expression of the CD34 gene in vascular endothelial cells. Blood. 1990; 15;75(12):2417-26.
8. Tineli RA Viaro F, Dalio MB, Reis GS, Bassetto S, Vicente WVA, et al. Mechanical forces and human saphenous vein: coronary artery bypass graft implications. Braz J Cardiovasc Surg. 2007;22(1):87-95.
FAPESP - The State of São Paulo Research Foundation - and FAEPA - The Teaching, Research and Assistence Foundation of the Hospital das Clínicas of the Medicine School in Ribeirão Preto, University of São Paulo






 All scientific articles published at bjcvs.org are licensed under a Creative Commons license
All scientific articles published at bjcvs.org are licensed under a Creative Commons license